- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
130 Good Manufacturing Practice (GMP) courses
SAM02: Regulatory Requirements and Guidance on Advertising and Promotion of Prescription Drugs in the USA
By Zenosis
In this course we explain how to advertise and promote prescription drugs in various media, whether to healthcare professionals or consumers, in compliance with legal requirements and guidance from the FDA.

SUB06: Variations to Marketing Authorisations in Europe
By Zenosis
Changes to the terms of marketing authorisations for medicinal products, called variations in Europe, must be notified to or approved by the relevant regulatory authorities. Variations include changes to the composition of products, their manufacturing processes, the way they are used, or the indications for which they are authorised. Common approaches are adopted within the European Economic Area to variations to marketing authorisations approved through the Centralised, Decentralised or Mutual Recognition Procedures. Recent legislation has substantially modified the regulatory requirements and extended them to purely national authorisations by member states. This module, which is fully up to date with the new legislation, covers the classification of variations into their several types and the regulatory requirements, guidance and procedures to be followed for each type.
ESS01: Essentials of EU and US Regulatory Affairs for Human Medicinal Products
By Zenosis
This foundation-level module is the ideal introduction for new entrants to the field of pharmaceutical regulatory affairs and compliance. It describes the principal requirements that must be satisfied to gain and maintain approval to market medicinal products in the USA and Europe. The legal framework and the roles of major players in regulation are presented. The life-cycle of a drug is outlined. The various procedures available for assessment and approval of products are described and their requirements outlined. Obligations to be fulfilled after marketing approval are discussed.

SUB03: The Mutual Recognition Procedure (MRP)
By Zenosis
his module describes the roles of the various players in the procedure, the sequence and duration of the stages involved, and the requirements on content, format and timing of submissions. It discusses the special issues that apply to generic products in the MRP.
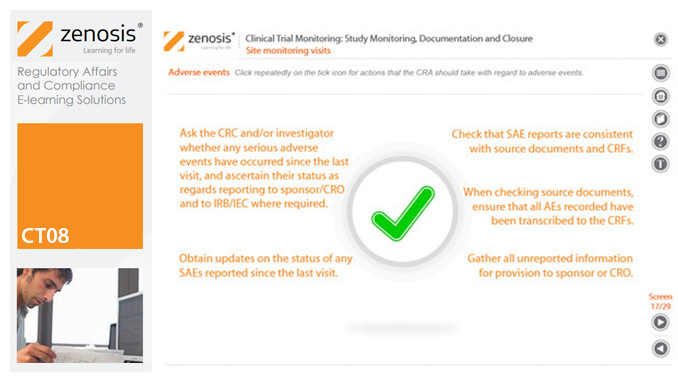
CT08: Clinical Trial Monitoring: Study Monitoring, Documentation and Closure
By Zenosis
The sponsor of a clinical trial must arrange for it to be monitored throughout its duration to ensure that the rights and wellbeing of subjects are protected, the trial data are accurate, complete and verified from source documents, and the conduct of the trial complies with the study protocol, Good Clinical Practice and regulatory requirements. In this module we describe how a Clinical Research Associate (CRA) monitors an ongoing trial to its conclusion.
SUB11: The Decentralised Procedure (DCP)
By Zenosis
This module describes the roles of the various players in the procedure, the sequence and duration of the stages involved, and the requirements on content, format and timing of submissions. It discusses the special issues that apply to generic products in the DCP.
SAM03: Consumer-directed Advertising and Online Promotion of Prescription Drugs in the USA
By Zenosis
Unlike the great majority of other countries, the advertising of prescription drugs directly to consumers is permitted in the USA. Spending by drug companies on direct-to-consumer (DTC) advertising of prescription drugs has increased more than four-fold over two decades, with a dramatic increase in the number of TV ads. In addition, the Internet and social media platforms have increasingly enabled companies to engage more actively with the public.

SAM01: Legal and Regulatory Framework for Advertising and Promotion of Prescription Drugs in the USA
By Zenosis
In this course we set out the legal framework for the regulation of advertising and promotion of prescription drugs in the USA. We identify the regulatory authorities and sources of guidance. We summarize basic requirements that advertisements and promotional labeling must meet, and we identify consequences that may follow failure to comply.

CT14: Clinical Trial Safety Reporting Requirements in the EU and USA
By Zenosis
This course sets out the legal and regulatory requirements for safety reporting in clinical trials of medicinal products under the jurisdictions of the European Union and the USA. It builds on the foundation laid by our companion course CT13, Safety Reporting in Clinical Trials, and provides greater detail of specific requirements in those jurisdictions.

CT13: Safety Reporting in Clinical Trials
By Zenosis
This course explains the regulatory requirements for the reporting of adverse events and suspected adverse reactions in clinical trials. It describes how investigators should report to sponsors, and how sponsors should report to regulatory authorities and other stakeholders in the safety of investigational products. It explains how events are characterized as serious or non-serious, expected or unexpected, and it distinguishes the requirements for each category. It describes controlled vocabularies used for coding of events in reports.
Search By Location
- Good Manufacturing Practice (GMP) Courses in London
- Good Manufacturing Practice (GMP) Courses in Birmingham
- Good Manufacturing Practice (GMP) Courses in Glasgow
- Good Manufacturing Practice (GMP) Courses in Liverpool
- Good Manufacturing Practice (GMP) Courses in Bristol
- Good Manufacturing Practice (GMP) Courses in Manchester
- Good Manufacturing Practice (GMP) Courses in Sheffield
- Good Manufacturing Practice (GMP) Courses in Leeds
- Good Manufacturing Practice (GMP) Courses in Edinburgh
- Good Manufacturing Practice (GMP) Courses in Leicester
- Good Manufacturing Practice (GMP) Courses in Coventry
- Good Manufacturing Practice (GMP) Courses in Bradford
- Good Manufacturing Practice (GMP) Courses in Cardiff
- Good Manufacturing Practice (GMP) Courses in Belfast
- Good Manufacturing Practice (GMP) Courses in Nottingham