- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
690 Manufacturing courses in Cardiff delivered On Demand
Diploma in Lean Leadership and Change Management
By iStudy UK
Lean Leadership allows you to transform your organisation's performance by changing its culture. The leader needs to change the way they are leading. The course is designed to teach you the strategies of lean leadership and guides you how to use culture for fostering business growth. The course helps you to understand the lean culture and lean leadership. The history of lean, the essential principles and practices, and the vital functions of the lean leader will be discussed in the course. You will also learn two ways of lean implementation: by redesigning the whole process or by acquiring the habits of teams and team leaders engaged in problem-solving and continuous improvement. Upon completion, you will be able to apply the lean culture in your company or workplace. What Will I Learn? Implement continuous improvement and lean culture (Toyota Production System). You will be able to eliminate waste and engage your team members in an on-going process of continuous improvement (kaizen). You will design a system of natural work and management teams who engage in continuous improvement. The student completing this course will know the essential problem solving methods employed in continuous improvement and will be able to lead his or her team in continuous improvement. Requirements There is no knowledge required to take this course other than a very fundamental understanding of the manager's job. Who is the target audience? Any manager, team leader, or anyone aspiring to improve the performance of his or her organization. Introduction Why FREE 00:04:00 What FREE 00:04:00 Curriculum FREE 00:05:00 How FREE 00:04:00 What is Culture 00:06:00 Culture Drivers 00:08:00 Resources 00:05:00 Activity 1 00:03:00 The House of Lean House of Lean 00:03:00 Evolution 00:04:00 Shingo 00:09:00 Honda 00:09:00 Activity 2 00:02:00 Lean Culture Principles & Practices JIT 00:07:00 Kanban 00:08:00 Continuous Improvement and Leadership 00:04:00 Waste Intro 00:10:00 Forms of Waste 1 00:06:00 Management Waste 00:07:00 Activity3 00:02:00 Scientific Method 00:07:00 Gemba Walk 00:06:00 Heijunka 00:05:00 Visual Display 00:07:00 Activity 4 00:02:00 Kaizen 00:06:00 The Job of the Lean Leader - To Challenge and To Serve Lean Leadership Intro 00:07:00 The Mind of the Lean Leader 00:04:00 The Mind of the Lean Leader - Transitions FREE 00:07:00 Lean Leadership1. External Challenge 00:06:00 Lean Leadership 2. Internal Challenge 00:06:00 Activity 5 00:02:00 Lead WSA 00:02:00 Be the Model 00:03:00 Coach Others 00:07:00 Motivation 00:04:00 Reinforce Your Strategy 00:04:00 Activity 6 00:01:00 Intrinsic Reinforcement 00:08:00 Getting to Lean - Designing the System for a New Culture Paths to Lean 00:05:00 How You Change 00:03:00 Design Thinking 00:08:00 Design Process 00:08:00 Capability Analysis 00:05:00 Charter 00:05:00 Activity 7 00:02:00 Design Team 00:06:00 Discovery 00:06:00 Discover the Process 00:07:00 Dream 00:07:00 Design technical 00:07:00 Design Structure 00:08:00 Social Design 00:06:00 Activity 8 00:03:00 VON Case Study 00:10:00 WSA Summary 00:04:00 Activity 9 00:02:00 The Habits of Continuous Improvement - The Team Kata Why Teams 00:06:00 Types of Teams 00:06:00 Team Charter 00:05:00 Team Maturity 00:05:00 Team Roles and Responsibilities 00:05:00 Team Agenda 00:05:00 Activity 10 00:02:00 Facilitation 1 00:06:00 Facilitation 2 00:04:00 Team Scoreboard 00:07:00 Activity 11 00:01:00 Map Team's Work 00:03:00 Attitudes 00:04:00 Five Whys 00:03:00 PDSA 1 00:04:00 PDSA 2 00:04:00 PDCA example 00:01:00 Activity 12 00:01:00 A3 00:06:00 Standard Work 00:05:00 Leader Standard Work 00:04:00 Activity 13 00:02:00 Summary 00:03:00 Additional Study Materials Additional Study Materials: Diploma in Lean Leadership and Change Management 00:00:00

Food isn’t just about taste — it’s a fine balance of science, safety, and smart processes. This course offers a deep dive into the essentials of food technology and the methods used to turn raw ingredients into the products found on supermarket shelves. You’ll explore how technology supports preservation, packaging, and processing — all while ensuring quality, hygiene, and industry standards are met. From microbial considerations to shelf-life strategies, every module is designed to sharpen your understanding of the sector’s inner workings. Ideal for those curious about what goes on behind the scenes in food manufacturing, this learning journey covers both traditional and contemporary techniques — without the fluff or fancy jargon. Whether you're aiming to enrich your knowledge for professional growth or simply fascinated by the mechanics of food production, this online course brings you the theory, terminology, and insights needed to confidently navigate the field. No apron required — just a good internet connection and a hunger for knowledge. Key Features CPD Accredited FREE PDF + Hardcopy certificate Fully online, interactive course Self-paced learning and laptop, tablet and smartphone-friendly 24/7 Learning Assistance Discounts on bulk purchases Course Curriculum of Food Technology and Food Processing Techniques Module 01 : Introduction to Food Technology Module 02 : Food Chemistry Module 03 : Food Biotechnology Module 04 : Food Microbiology Module 05 : Food Processing Techniques Part-1 Module 06 : Food Processing Techniques Part-2 Module 07 : Food Packaging and Sustainability Learning Outcomes: Analyze food compositions for optimal processing efficiency. Apply biotechnological methods for food production enhancement. Evaluate microbial impacts on food safety and preservation techniques. Demonstrate proficiency in various food processing techniques. Implement sustainable packaging solutions in food industry practices. Comprehend the broader implications of food technology on environmental sustainability. Accreditation This course is CPD Quality Standards (CPD QS) accredited, providing you with up-to-date skills and knowledge and helping you to become more competent and effective in your chosen field. CPD 10 CPD hours / points Accredited by CPD Quality Standards Who is this course for? Professionals transitioning to remote work. Individuals seeking to enhance virtual collaboration skills. Remote employees looking to boost productivity. Freelancers and independent contractors. Managers overseeing remote teams. Students preparing for a virtual work landscape. Entrepreneurs building virtual businesses. Anyone eager to excel in remote work scenarios. Career path Remote Team Manager Virtual Project Coordinator Freelance Consultant Online Business Owner Virtual Communication Specialist Remote Productivity Coach Certificates Digital certificate Digital certificate - Included Also, you can have your FREE printed certificate delivered by post (shipping cost £3.99 in the UK). Hard copy certificate Hard copy certificate - Included Also, you can have your FREE printed certificate delivered by post (shipping cost £3.99 in the UK). For all international addresses outside of the United Kingdom, the delivery fee for a hardcopy certificate will be only £10. Our certifications have no expiry dates, although we do recommend that you renew them every 12 months.

Chemical hygiene isn’t just about keeping a lab coat clean—it’s about staying safe while working with substances that don’t take kindly to guesswork. This Chemical Hygiene and Engineering Course blends sharp knowledge with safety insight, helping you understand everything from chemical handling protocols to ventilation systems and control measures. You’ll explore how chemical reactions work, what containment means in engineering systems, and why even a small slip in hygiene can lead to a big bang—literally. Whether you’re curious about safe lab setups or how chemical exposure limits are determined, this course keeps the science straightforward and the safety front and centre. With engineering principles guiding chemical safety strategies, this course teaches you how proper design reduces risk. Learn about exposure control plans, risk assessments, chemical storage systems, and air quality engineering—all from the comfort of your screen, no gloves required. It’s ideal for those working in labs, manufacturing, or anywhere that safety goggles are more than just a fashion statement. You’ll leave with knowledge that sticks—without touching a Bunsen burner. Let’s face it, chemicals don’t do well with shortcuts, and neither should your understanding of them. Key Features CPD Accredited FREE PDF + Hardcopy certificate Fully online, interactive course Self-paced learning and laptop, tablet and smartphone-friendly 24/7 Learning Assistance Discounts on bulk purchases Course Curriculum Module 01: Introduction to Chemical Hygiene and Engineering Module 02: Chemical Hazards and Toxicology Module 03: Chemical Storage and Handling Module 04: Personal Protective Equipment (PPE) and Engineering Controls Module 05: Laboratory Safety and Best Practices Module 06: Chemical Process Safety Learning Outcomes: Identify chemical hazards and understand their toxicological implications. Implement effective chemical storage and handling practices in the workplace. Choose and use personal protective equipment (PPE) with precision. Apply engineering controls to enhance safety in diverse work settings. Demonstrate best practices for laboratory safety and operations. Safeguard against potential risks through a deep understanding of chemical process safety. Accreditation This course is CPD Quality Standards (CPD QS) accredited, providing you with up-to-date skills and knowledge and helping you to become more competent and effective in your chosen field. Certificate After completing this course, you will get a FREE Digital Certificate from Training Express. CPD 10 CPD hours / points Accredited by CPD Quality Standards Who is this course for? Laboratory Technicians Chemical Engineers Safety Officers Researchers Environmental Scientists Facility Managers Process Engineers Occupational Health Specialists Career path Chemical Safety Officer Laboratory Manager Process Engineer Environmental Health and Safety Specialist Research Scientist Occupational Health Advisor Certificates Digital certificate Digital certificate - Included Once you've successfully completed your course, you will immediately be sent a FREE digital certificate. Hard copy certificate Hard copy certificate - Included Also, you can have your FREE printed certificate delivered by post (shipping cost £3.99 in the UK). For all international addresses outside of the United Kingdom, the delivery fee for a hardcopy certificate will be only £10. Our certifications have no expiry dates, although we do recommend that you renew them every 12 months.

Safety showers and eye wash stations are critical in ensuring the well-being of individuals working with hazardous materials. These emergency safety equipment items are designed to provide immediate decontamination in case of accidental exposure to harmful chemicals or substances. By having these systems in place, workplaces significantly reduce the risk of injury and facilitate swift responses in critical situations. This course aims to provide learners with essential knowledge on the correct installation, maintenance, and use of safety showers and eye wash stations, ensuring that workplace environments remain safe for all employees. The course offers in-depth insights into the regulatory standards and guidelines that govern these safety systems. It addresses the importance of regular checks, prompt maintenance, and the correct usage of safety showers and eye wash stations during emergencies. Participants will learn the specifications for their setup, the types of hazards they protect against, and how to ensure proper functionality in case of urgent need. Whether you're in manufacturing, laboratories, or any environment that involves exposure to hazardous substances, this course will equip you with the necessary information to safeguard the health of individuals on your site. Key Features CPD Accredited FREE PDF + Hardcopy certificate Fully online, interactive course Self-paced learning and laptop, tablet and smartphone-friendly 24/7 Learning Assistance Discounts on bulk purchases Course Curriculum Module 01: Introduction to Safety Showers and Eye Washes Module 02: Operating Procedures Module 03: Proper Utilisation Module 04: First Aid for Chemical Injuries Module 05: Maintenance and Testing Module 06: Emergency Procedures and Preparedness Module 07: Safety Culture and Best Practices Learning Outcomes: Implement proper safety shower and eye wash procedures for workplace incidents. Demonstrate first aid skills tailored to chemical injuries for immediate response. Execute maintenance and testing routines for optimal safety equipment functionality. Execute emergency procedures, ensuring a swift and effective response to incidents. Foster a safety culture, integrating best practices into daily operations. Demonstrate expertise in safety protocols to enhance workplace well-being. Accreditation This course is CPD Quality Standards (CPD QS) accredited, providing you with up-to-date skills and knowledge and helping you to become more competent and effective in your chosen field. Certificate After completing this course, you will get a FREE Digital Certificate from Training Express. CPD 10 CPD hours / points Accredited by CPD Quality Standards Who is this course for? Laboratory Technicians Industrial Workers Safety Officers Environmental Health Specialists Facility Maintenance Staff Chemical Plant Operators Emergency Response Teams Health and Safety Coordinators Career path Industrial Safety Officer Environmental Health Specialist Chemical Plant Operator Health and Safety Coordinator Emergency Response Team Member Laboratory Safety Technician Certificates Digital certificate Digital certificate - Included Once you've successfully completed your course, you will immediately be sent a FREE digital certificate. Hard copy certificate Hard copy certificate - Included Also, you can have your FREE printed certificate delivered by post (shipping cost £3.99 in the UK). For all international addresses outside of the United Kingdom, the delivery fee for a hardcopy certificate will be only £10. Our certifications have no expiry dates, although we do recommend that you renew them every 12 months.

The hand, wrist, and fingers are remarkable parts of the body that perform countless tasks every day, yet they are often vulnerable to injuries and strains. This course offers a thorough understanding of the anatomy and physiology of these vital components, helping learners recognise common disorders and injuries that may affect them. With an emphasis on prevention, the training explores effective methods to reduce ergonomic risks and maintain long-term health in various settings. Beyond theory, the course highlights essential safety measures, including correct tool usage and maintenance, alongside the importance of personal protective equipment (PPE) designed specifically for hand safety. Whether you work in an office environment, manufacturing, or other industries, this training provides valuable knowledge to safeguard your hands and minimise injury risk. Designed for easy learning without the need for physical attendance, it’s a smart investment in wellbeing and productivity.. Key Features CPD Accredited FREE PDF + Hardcopy certificate Fully online, interactive course Self-paced learning and laptop, tablet and smartphone-friendly 24/7 Learning Assistance Discounts on bulk purchases Course Curriculum of Hand, Wrist & Finger Safety Training Lesson 01 :Physiology and Anatomy of Hand, Wrist and Finger Lesson 02 :Hand, Wrist & Finger Disorders Lesson 03 :Common Injuries and First Aid Lesson 04 :Preventing Ergonomic Injuries Lesson 05 :Proper Techniques & Practices Lesson 06: Proper Use and Maintenance of Tools Lesson 07:Personal Protective Equipment (PPE) for Hand Safety Learning Outcomes: Analyze the Physiology and Anatomy of the Hand, Wrist, and Finger for enhanced safety awareness. Identify and address Hand, Wrist & Finger Disorders for a healthier workplace environment. Administer effective First Aid for common Hand, Wrist, and Finger injuries, ensuring prompt care. Implement ergonomic principles to Prevent Ergonomic Injuries and promote worker well-being. Demonstrate mastery in Proper Techniques & Practices for optimal Hand Safety. Proficiently employ and maintain Tools, guaranteeing their safe and efficient use. Accreditation This course is CPD Quality Standards (CPD QS) accredited, providing you with up-to-date skills and knowledge and helping you to become more competent and effective in your chosen field. CPD 10 CPD hours / points Accredited by CPD Quality Standards Who is this course for? Health and Safety Professionals seeking specialized knowledge. Occupational Health Practitioners focusing on hand safety. Workers in industries with a high risk of hand injuries. Employers looking to enhance workplace safety measures. Individuals aspiring to pursue a career in Occupational Health and Safety. Students studying Health, Safety, and Environment (HSE) disciplines. Industrial Hygienists dedicated to hand safety. Risk Management Specialists prioritizing hand protection. Career path Health and Safety Officer Occupational Health Advisor Ergonomics Consultant Industrial Hygienist Risk Management Analyst Safety Compliance Officer Certificates Digital certificate Digital certificate - Included Once you've successfully completed your course, you will immediately be sent a FREE digital certificate. Hard copy certificate Hard copy certificate - Included Also, you can have your FREE printed certificate delivered by post (shipping cost £3.99 in the UK). For all international addresses outside of the United Kingdom, the delivery fee for a hardcopy certificate will be only £10. Our certifications have no expiry dates, although we do recommend that you renew them every 12 months.
Lean Tools and Processes: How to Use Them in Your Business
By IIL Europe Ltd
Lean Tools and Processes: How to Use Them in Your Business This presentation provides an overview of the most common, traditional Lean tools and methods that can be applied to any business -- from the shop floor to the back office, and from retail businesses to service industries. Topics covered will include Standard Work, 5S, Error Proofing, and others. The required culture change within a company and adoption of a "Lean Mindset" will also be discussed. This and other IIL Learning in Minutes presentations qualify for PDUs. Some titles, such as Agile-related topics may qualify for other continuing education credits such as SEUs, or CEUs. Each professional development activity yields one PDU for one hour spent engaged in the activity. Some limitations apply and can be found in the Ways to Earn PDUs section that discusses PDU activities and associated policies. Fractions of PDUs may also be reported. The smallest increment of a PDU that can be reported is 0.25. This means that if you spent 15 minutes participating in a qualifying PDU activity, you may report 0.25 PDU. If you spend 30 minutes in a qualifying PDU activity, you may report 0.50 PDU.

HACCP: HACCP Course Online Unlock the Power of HACCP: HACCP Course: Enrol Now! The European Regulation (EC) No 852/2004 on Food Hygiene is a legal requirement for catering, manufacturing, and retail businesses. In addition, the Hazard Analysis and Critical Control Point (HACCP) system is a food safety management system required by all businesses that serve food and beverages, including hotels, restaurants, and hospitals. This HACCP: HACCP Course is designed to introduce you to food hygiene legislation as well as the seven HACCP principles. You will also learn how to implement a HACCP system in the workplace, putting key safety control measures into action. Learning Outcomes After completing this HACCP: HACCP Course, you will be able to: Describe the fundamentals of HACCP. Explain Microbial Hazards. Gain a solid understanding of Food Safety hazards. Describe Prerequisite Programmes. Create a HACCP system. Describe hazard analysis and control measures. By the end of this HACCP: HACCP Course, you will have a solid understanding of food safety management fundamentals, including preventing and identifying food safety hazards, maintaining an excellent standard of service. Also, a healthy working environment for all. Courses you will get: Course 01: Level 3 HACCP Training Course Course 02: Level 2 Food Hygiene and Safety For Catering Course 03: Level 2 Food Hygiene and Safety Course 04: Level 2 HACCP Training Course For Catering & Retail [ Note: Free PDF certificate as soon as completing the HACCP: HACCP course] HACCP: HACCP Course Online This HACCP: HACCP Course consists of 15 modules. Course Curriculum of HACCP: HACCP Course Module 01: An Overview of HACCP Module 02: Laws Related to HACCP Module 03: HACCP Alternatives Module 04: Planning a HACCP System Module 05: Understanding Food Safety Hazards Module 06: Prerequisite Programmes Module 07: Creating the HACCP System Module 08: Principle 1: Hazard Analysis Module 09: Principle 2: Critical Control Points Module 10: Principle 3: Critical Limits Module 11: Principle 4: Monitoring Critical Control Points Module 12: Principle 5: Corrective Action Module 13: Principle 6: Verification of the HACCP System Module 14: Principle 7: Documentation Module 15: Implementing the HACCP System Assessment Method of HACCP: HACCP Course After completing HACCP: HACCP Course, you will get quizzes to assess your learning. You will do the later modules upon getting 60% marks on the quiz test. Apart from this, you do not need to sit for any other assessments. Certification of HACCP: HACCP Course After completing the HACCP: HACCP course, you can instantly download your certificate for FREE. The hard copy of the certification will also be delivered to your doorstep via post, which will cost £13.99. Who is this course for? HACCP: HACCP Course Online This HACCP: HACCP Course is ideal for those who work in: - Restaurants or cafes., Hospitality and catering., Kitchens in schools and workplaces., Stalls, selling food., Supermarkets and butchers. Requirements HACCP: HACCP Course Online To enrol in this HACCP: HACCP Course, students must fulfil the following requirements: Good Command over English language is mandatory to enrol in our HACCP: HACCP Course. Be energetic and self-motivated to complete our HACCP: HACCP Course for Catering Course. Basic computer Skill is required to complete our HACCP: HACCP Course for Catering Course. If you want to enrol in our HACCP: HACCP Course, you must be at least 15 years old. Career path HACCP: HACCP A formal HACCP: HACCP qualification ensures that you, as an employee, have up to date knowledge of food management systems and can demonstrate an excellent understanding of food hygiene and safety regulations in the workplace. This course can be applied to the following: Food and restaurant business Catering and hospitality School/work kitchens Fast food industry Bars and pubs Hotels

Complete Project Management, Operations Management, Business Management & analysis Level 7
5.0(3)By School Of Health Care
Operations Management: Operations Management Course Online Introducing Operations Management: Operations Management Course "Empowering the Next Generation" Online course! Operations Management: Operations Management Course is a fast-paced field with a clear goal. For those in operations management: operations management, the goal is to maximize profits by balancing costs and revenues. By incorporating human resources, raw materials, technology, and equipment across the board, Operations Management: Operations Management aims to administer business practices in a way that drives efficiency and superior net profits. The Operations Management: Operations Management Course prepares learners to succeed in their professional lives by teaching them the concepts of Supply Chain Management and its fundamentals, Risk Management and Time Management, Talent Management, Project Management, and Performance Management, Meeting Management, Knowledge Management, and Negotiation skills. Main Course: Level 7 Diploma in Operations Management Course Free Courses included with Operations Management: Operations Management Course: Course 01: Level 7 Project Management Course 02: Level 7 Diploma in Facilities Management Course 03: Level 5 Diploma in Business Analysis Course 04: Level 2 Certificate in Business Management Special Offers of this Operations Management: Operations Management Course: This Operations Management: Operations Management Course includes a FREE PDF Certificate. Lifetime access to this Operations Management: Operations Management Course Instant access to this Operations Management: Operations Management Course 24/7 Support Available to this Operations Management: Operations Management Course [ Note: Free PDF certificate as soon as completing the Operations Management: Operations Management course] Operations Management: Operations Management Course Online This Operations Management: Operations Management Course consists of 20 modules. Course Curriculum of Level 7 Operations Management Module 01: Understanding Operations Management Module 02: Understanding Process Management Module 03: Understanding Supply Chain Management Module 04: Understanding Planning & Sourcing Operations Module 05: Understanding Talent Management Module 06: Understanding Procurement Operations Module 07: Understanding Manufacturing and Service Operations Module 08: Understanding Succession Planning Module 09: Understanding Project Management Module 10: Understanding Quality Control Module 11: Understanding Product and Service Innovation Module 12: Understanding Communication Skills Module 13: Understanding Negotiation Techniques Module 14: Understanding Change Management Module 15: Understanding Maintenance Management Module 16: Understanding Conflict Management Module 17: Understanding Stress Management Module 18: Understanding Business Ethics for the Office Module 19: Understanding Business Etiquette Module 20: Understanding Risk Management Assessment Method of Operations Management: Operations Management Course After completing Operations Management: Operations Management Course, you will get quizzes to assess your learning. You will do the later modules upon getting 60% marks on the quiz test. Certification of Operations Management: Operations Management Course After completing the Operations Management: Operations Management Course, you can instantly download your certificate for FREE. Who is this course for? Operations Management: Operations Management Course Online This Operations Management: Operations Management Course is suitable for anyone passionate and ambitious about Operations Management: Operations Management. Learners can take this Operations Management: Operations Management Course even if they have no prior knowledge of Operations Management: Operations Management, and there are no age restrictions. Requirements Operations Management: Operations Management Course Online To enrol in this Operations Management: Operations Management Course, students must fulfil the following requirements: Good Command over English language is mandatory to enrol in our Operations Management: Operations Management Course. Be energetic and self-motivated to complete our Operations Management: Operations Management Course. Basic computer Skill is required to complete our Operations Management: Operations Management Course. If you want to enrol in our Operations Management: Operations Management Course, you must be at least 15 years old. Career path Operations Management: Operations Management Course This Operations Management: Operations Management could be extremely beneficial to business owners and aspiring entrepreneurs to pursue their goals. The following are more formal positions in the field of operations management: Operations Manager Operations Analyst Operations Officer S.C Manager Inventory Manager Supply Services Representative

Advanced Innovations in Food Technology Course
By One Education
Food science is no longer confined to the pages of a textbook — it's a dynamic field constantly reshaped by innovation, technology, and a dash of unexpected brilliance. The Advanced Innovations in Food Technology Course is designed for forward-thinkers eager to stay one step ahead in an industry where yesterday’s methods are quickly left on the shelf. From AI in food safety to smart packaging and sustainable production, this course explores what’s next in food tech — and why it matters now more than ever. Whether you're working in food manufacturing, research, or quality control, or simply fascinated by what tomorrow's plate might look like, this course dishes out insightful knowledge served with precision. With a keen focus on the cutting-edge without veering into fantasy, the content is both grounded and future-focused. No fluff, no filler — just thought-provoking material for professionals hungry for knowledge that actually feeds progress. Key Features The Advanced Innovations in Food Technology course includes the following offers: CPD Quality Standards Accredited Advanced Innovations in Food Technology Course Lifetime Access No Hidden Fees 24/7 Tutor Support Certificate Available with reasonable price Discover a wealth of learning opportunities with our diverse range of online courses including Advanced Innovations in Food Technology. From professional development to personal enrichment, we offer courses in a variety of subjects including business, technology, arts, languages, and more. This Advanced Innovations in Food Technology course is carefully curated by industry experts and delivered through engaging multimedia content, interactive quizzes, and hands-on projects to ensure an immersive learning experience. Curriculum Module 1 Introduction To Food Technology Module 2 Food Chemistry Module 3 Food Biotechnology Module 4 Food Microbiology Module 5 Food Processing Techniques Part-1 Module 6 Food Processing Techniques Part-2 Module 7 Food Packaging And Sustainability Why This Online Course is Better for You: Unlike traditional classroom settings, our online Advanced Innovations in Food Technology course offers flexibility and convenience tailored to fit your busy lifestyle. With 24/7 access to Advanced Innovations in Food Technology course materials and the ability to learn at your own pace, you can study whenever and wherever it's most convenient for you. Plus, our online platform provides a supportive community of fellow learners and dedicated instructors to guide you every step of the way. CPD 10 CPD hours / points Accredited by CPD Quality Standards Who is this course for? Our Advanced Innovations in Food Technology course is designed for anyone eager to expand their knowledge and skills, regardless of their background or experience level. Whether you're a working professional looking to upskill, a student seeking supplemental education, or an enthusiast pursuing a passion project, our courses are accessible to learners of all ages and abilities. Career path Invest in your future and unlock new career opportunities with our online Advanced Innovations in Food Technology course. Gain valuable skills and credentials that are in high demand in today's competitive job market. Whether you're looking to switch careers, climb the corporate ladder, or start your own business, This course provide the expertise and certification you need to succeed. Certificates Digital certificate Digital certificate - £9 Hard copy certificate Hard copy certificate - £15 A Hardcopy Certificate (international): £15 + £10 (postal charge) = £25

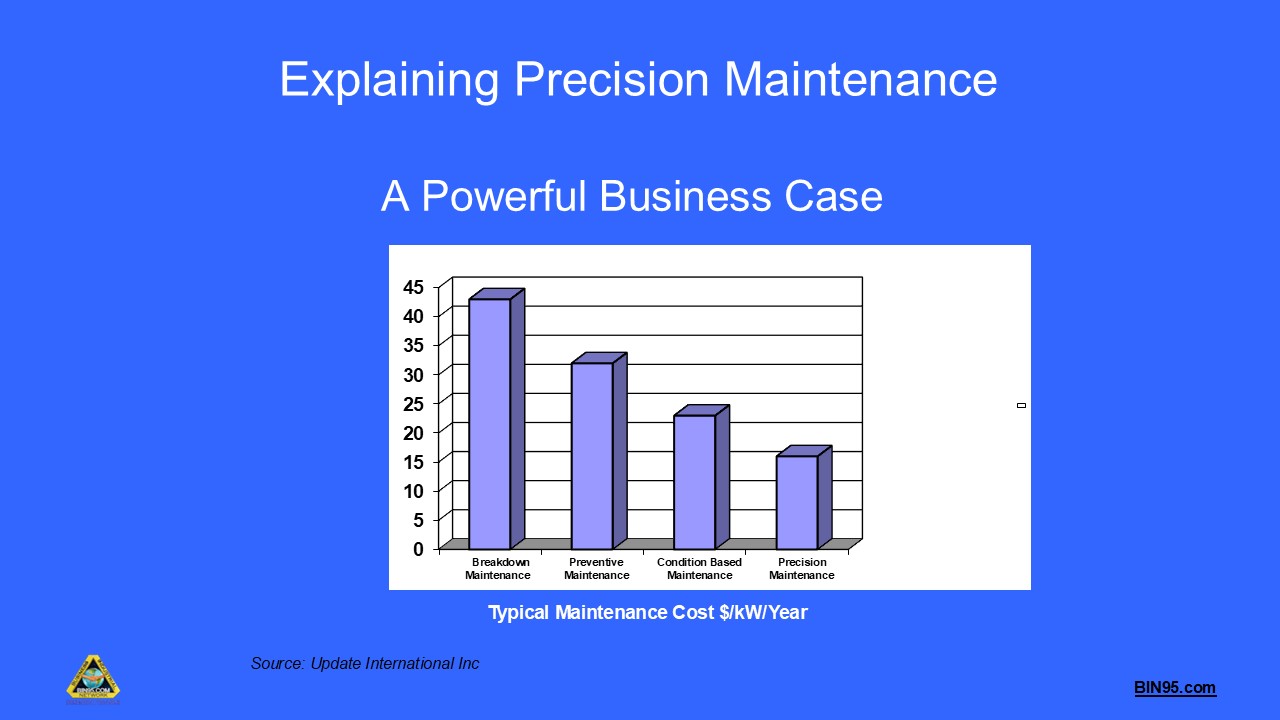
Precision Maintenance Training
By BIN95
This free course, provided by the BIN95 Manufacturing Training Division, provides a comprehensive understanding of precision maintenance as a critical component of world-class rotating machinery reliability. Participants will learn the business case for precision maintenance, the standards and best practices required, and the practical steps to implement precision maintenance in industrial environments. The course covers key topics, including vibration analysis, alignment, balancing, lubrication, fit and tolerance, torque standards, and the development of a precision maintenance culture. Through real-world examples, standards references (ISO, ANSI), and practical procedures, learners will gain the skills to reduce maintenance costs, increase equipment reliability, and drive continuous improvement in plant operations. Ideal for maintenance professionals, engineers, and reliability managers seeking to elevate their maintenance practices to world-class standards.