- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
Certified Associate in Project Management (CAPM) Exam Prep: In-House Training
By IIL Europe Ltd
Certified Associate in Project Management (CAPM)® Exam Prep: In-House Training: In-House Training This course gives you the knowledge you need to pass the exam and covers CAPM®-critical information on project management theory, principles, techniques, and methods Are you planning on taking the CAPM® examination? This course gives you the knowledge you need to pass the exam and covers CAPM®-critical information on project management theory, principles, techniques, and methods. You'll also have an opportunity for practical applications and time to review the kinds of questions you'll find in the CAPM® Exam. What you Will Learn Apply for the CAPM® Examination Develop a personal exam preparation plan Describe the structure, intent, and framework principles of the current edition of the PMBOK® Guide Explain the PMBOK® Guide Knowledge Areas, as well as their inter-relationships with the each other and the Process Groups Getting Started Program orientation The CAPM® certification process Certified Associate in Project Management (CAPM®) Examination Content Outline CAPM® eligibility requirements Code of Ethics and Professional Conduct Application options Foundation Concepts Skills and qualities of a project manager Project management terminology and definitions Relationship of project, program, portfolio, and operations management Project lifecycle approaches Project Integration Management Review Project Integration Management Knowledge Area Develop Project Charter Develop Project Management Plan Direct and Manage Project Work Manage Project Knowledge Monitoring and Controlling Perform Integrated Change Control Close Project or Phase Project Stakeholder Management Review Project Stakeholder Management Knowledge Area Identify Stakeholders Plan Stakeholder Engagement Manage Stakeholder Engagement Monitor Stakeholder Engagement Project Scope Management Review Project Scope Management Knowledge Area Plan Scope Management Collect Requirements Define Scope Create WBS Validate Scope Control Scope Project Schedule Management Review Project Schedule Management Knowledge Area Plan Schedule Management Define Activities Sequence Activities Estimate Activity Durations Develop Schedule Control Schedule Project Cost Management Review Project Cost Management Knowledge Area Plan Cost Management Estimate Costs Determine Budget Control Schedule Project Resource Management Review Project Resource Management Knowledge Area Plan Resource Management Estimate Activity Resources Acquire Resources Develop Team Manage Team Control Resources Project Quality Management Review Project Quality Management Knowledge Area Plan Quality Management Manage Quality Control Quality Project Risk Management Review Project Risk Management Knowledge Area Plan Risk Management Identify Risks Perform Qualitative Risk Analysis Perform Quantitative Risk Analysis Plan Risk Responses Implement Risk Responses Monitor Risks Project Communications Management Review Project Communications Management Knowledge Area Plan Communications Management Manage Communications Monitor Communications Project Procurement Management Review Project Procurement Management Knowledge Area Plan Procurement Management Conduct Procurements Control Procurements Summary and Next Steps Program Review Mock CAPM® Exam Getting Prepared for the CAPM® Exam After the CAPM® Exam

Effecting Business Process Improvement: In-House Training
By IIL Europe Ltd
Effecting Business Process Improvement: In-House Training Business analysts facilitate the solution of business problems. The solutions are put into practice as changes to the way people perform in their organizations and the tools they use. The business analyst is a change agent who must understand the basic principles of quality management. This course covers the key role that business analysts play in organizational change management. What you will Learn You will learn how to: Define and document a business process Work with various business modeling techniques Perform an enterprise analysis in preparation for determining requirements Analyze business processes to discern problems Foundation Concepts Overview of business analysis and process improvement Defining the business process Introducing the proactive business analyst Focusing on business process improvement for business analysts Launching a Successful Business Process Improvement Project Overview of the launch phase Understanding and creating organizational strategy Selecting the target process Aligning the business process improvement project's goals and objectives with organizational strategy Defining the Current Process Overview of current process phase Documenting the business process Business modeling options: work-flow models Business modeling options: Unified Modeling Language (UML) model adaptations for business processes Analyzing the Current Process Process analysis overview Evaluation: establishing the control group Opportunity techniques: multi-discipline problem-solving Opportunity techniques: matrices Building and Sustaining a Recommended Process Overview of the recommended process and beyond Impact analysis Recommended process Transition to the business case Return to proactive state

Risk Management for IT Projects: In-House Training
By IIL Europe Ltd
Risk Management for IT Projects: In-House Training IT projects may have direct bottom-line impact on the organization, cost millions of dollars, cause organizational change and change the way the organization is perceived by clients. Many IT projects are notoriously hard to predict and are filled with risk. IT Risk Management takes a comprehensive look at IT project risk management using PMI's PMBOK® Guide Risk Management Model in the context of IT Project Life Cycle phases. The goal of this course is to arm the practitioner with a rigorous, common-sense approach to addressing uncertainty in projects. This approach includes the ability to influence project outcomes, avoid many potential project risks, and be ready to calmly and efficiently respond to unavoidable challenges. What you will Learn You'll learn how to: Describe the risk management process, using the PMBOK® Guide's standard models and terminology Discuss the potential barriers to managing risk effectively in IT project organizations Develop an effective risk management plan for IT projects Identify project risks using IT-specific, practical tools Analyze individual risk events and overall project risk using IT-specific, practical approaches Plan effective responses to IT-specific risk based on the results of risk analysis and integrate risk responses into project schedules and cost estimates Manage and control risk throughout the IT project life cycle Implement selected elements of IT project risk management on your next project Foundation Concepts Basic concepts and purpose Risk and project constraints Risk and corporate cultures Risk management and IT PLC standards Plan Risk Management for IT Projects Plan Risk management process Plan Risk management activities Design a standard template Assess the project-specific needs Tailor the template Produce a project-specific risk management plan Gain consensus and submit as part of overall project plan A risk management plan of IT projects Identify Risks for IT Projects Identify risk process overview Risk categories and examples Risk identification tools Risk events by project life-cycle phases Perform Risk Analysis for IT Projects Perform qualitative risk analysis overview Core qualitative tools for IT projects Auxiliary qualitative tools for cost and schedule estimates When to use quantitative analysis for IT projects Plan Risk Response for IT Projects Plan risk response overview Active risk response strategies for IT projects (Threat and Opportunity) Acceptance and contingency reserves Contingency planning for IT projects Plan risk responses for IT projects Implement Risk Response for IT Projects Implement Risk Responses Executing Risk Response Plans Techniques and Tools Used Continuous Risk Management Monitor Risks for IT Projects Monitor risks overview Monitor risks tips for IT projects Technical performance measurement systems Risk management implementation for IT projects

Good Clinical Practice Auditing - Principles and Practice
By Research Quality Association
Course Information Our comprehensive course is used as a gateway to those stepping into the world of auditing clinical studies. Tailored for those already acquainted with Good Clinical Practice (GCP) and those transitioning from other audit disciplines, this programme stands as a pivotal guide. Pre-existing knowledge of GCP will significantly enhance your learning experience in auditing against these guidelines. How is this course run? Engage in immersive workshops providing hands-on practice with auditing techniques in a GCP context. Our seasoned tutors, boasting extensive audit experience, intertwine theory with practical insights drawn from their own professional journeys. What will I learn? A comprehensive understanding of the historical backdrop and objectives driving Good Clinical Practice, incorporating the latest industry developments Solid grounding in quality assurance activities aligned with regulatory standards Insight into potential pitfalls within clinical trials and the pivotal role of auditors in addressing these issues Clarity on the roles and responsibilities inherent to clinical trials auditing Exposure to a diverse range of audit techniques complemented by illustrative examples and supportive documents A nuanced understanding of regulatory inspectors' activities Expanded professional networks to propel your auditing career forward. Benefits include: A clear understanding of the role of the auditor under Good Clinical Practice improved audits Improved Good Clinical Practice compliance for your clinical trials. This course is structured to encourage delegates to: Discuss and develop ideas Solve specific problems Examine particular aspects of Good Clinical Practice. Tutors Tutors will be comprised of (click the photos for biographies): Rosemarie Corrigan EVP Global Quality, Worldwide Clinical Trials Cathy Dove Director and Owner, Dove Quality Solutions Julie Kelly Associate Director, Clinical Quality Assurance, Corcept Therapeutics Susana Tavares Director of Research Quality Assurance, - Programme Please note timings may be subject to alteration. Day 1 12:30 Registration 13:00 Welcome and Objectives for the first day of the course 13:30 Laying the Foundations Introduction to the clinical development process, the concepts of quality assurance, quality control and audit. 14:30 Break 15:00 Patient Protection Requirements for informed consent and ethics committee. Access to source documentation. Including a patient protection exercise. 16:05 Workshop 1 - Case Study on Informed Consent 16:45 End of Day Questions and Answers 17:00 Close of Day Day 2 08:50 Questions and Answers from Day 1 09:00 Effective Site Audits The procedures involved in selecting and setting up audits at investigator sites. 09:40 Workshop 1 - Planning the Effective Audit 10:30 Break 10:45 Source Data Verification The need for and purpose of verifying data. 11:25 Workshop 2 - Source Data Verification 12:30 Lunch 13:30 IMP Management The requirements surrounding the distribution of investigational medicinal products. Accountability from release to destruction. 14:15 Critical Document Audits The conduct of other study specific audits including protocols, databases and reports. 15:00 Break 15:15 Non-compliance Determining the acceptability of data. 16:00 Fraud - Fact or Fiction? How to identify fraud and its consequences 16:45 End of Day Questions and Answers 17:00 Close of Day Day 3 08:50 Questions and Answers from Days 1 and 2 09:00 Auditing Third Parties A review of audits of contract research organisations. 10:00 System Audits The concept of auditing processes across many clinical trials, including a practical exercise in process mapping. 10:45 Break 11:00 Workshop 3 - Process Mapping 11:45 Effective Audits Where theory meets reality. 12:30 Lunch 13:20 Audit Reports - Closing the Loop An examination of the processes which follow the evidence gathering phase of the audit. 14:20 Workshop 4 - Audit Reports Audit reports, corrective and preventive action. 15:00 Break 15:10 Regulatory Inspection Auditors and regulatory inspections -how the QA team can help the organisation to perform during a regulatory inspection. 15:55 Final Questions and Answers 16:10 Close of Course Extra Information Face-to-face course Course Material Course material will be available in PDF format for delegates attending this course. The advantages of this include: Ability for delegates to keep material on a mobile device Ability to review material at any time pre and post course Environmental benefits – less paper being used per course. The material will be emailed in advance of the course and RQA will not be providing any printed copies of the course notes during the training itself. Delegates wishing to have a hard copy of the notes should print these in advance to bring with them. Alternatively delegates are welcome to bring along their own portable devices to view the material during the training sessions. Remote course Course Material This course will be run completely online. You will receive an email with a link to our online system, which will house your licensed course materials and access to the remote event. Please note this course will run in UK timezone. The advantages of this include: Ability for delegates to keep material on a mobile device Ability to review material at any time pre and post course Environmental benefits – less paper being used per course Access to an online course group to enhance networking. You will need a stable internet connection, a microphone and a webcam. CPD Points 17 Points Development Level Develop

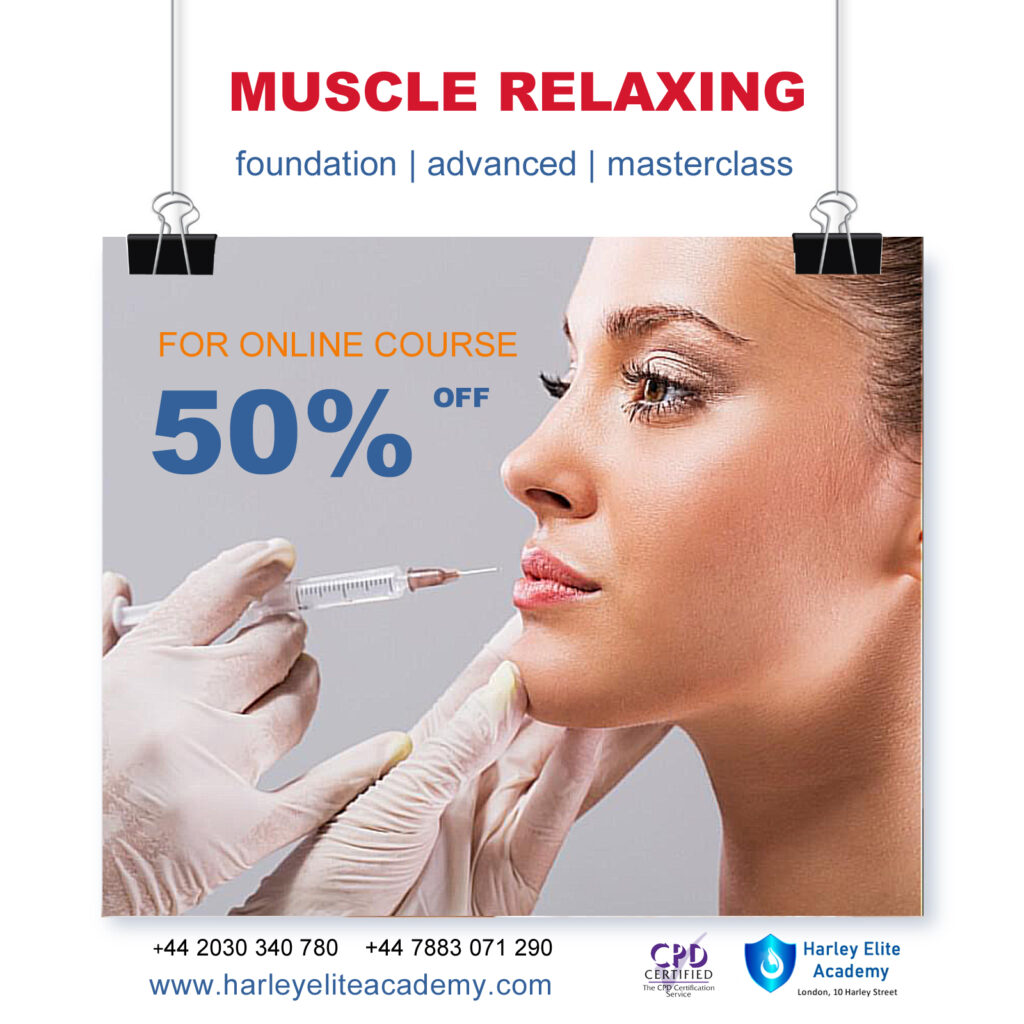
MUSCLE RELAXING | BOTOX®
By Harley Elite Academy (HeLa)
Foundation • Advanced • Masterclass 8 CPD POINTS 1 DAY INTENSIVE COURSE ONLINE or IN-CLINIC NOTE! After booking we will contact you for scheduling the exact course date! Courses dates are subject to change due to mentors availability. We will inform you via email if a date becomes available! You need to be medically qualified as a doctor, dentist, nurse, pharmacist or paramedic with full governing body registration and have completed a Foundation Filler Course and to have administered a number of cases. Additional information ATTENDANCE ONLINE (theory), IN-CLINIC (Practice) COURSE LEVEL BEGINNER | Foundation Course, INTERMEDIATE | Advanced Course, EXPERT | Masterclass Course
Các Mẫu Giày Tây Nam Cao Cấp Phù Hợp Cho Môi Trường Công Sở
By Các Mẫu Giày Tây Nam Cao Cấp Phù Hợp Cho Môi Trường Công Sở
Các Mẫu Giày Tây Nam Cao Cấp Phù Hợp Cho Môi Trường Công Sở Trong môi trường công sở, trang phục không chỉ thể hiện sự chuyên nghiệp mà còn tạo ấn tượng đầu tiên mạnh mẽ với đối tác và đồng nghiệp. Một phần quan trọng không thể thiếu của bộ trang phục công sở chính là giày tây. Giày tây nam không chỉ giúp bạn hoàn thiện vẻ ngoài lịch lãm mà còn phản ánh phong cách và sự tôn trọng quy tắc thời trang. Hôm nay, chúng ta sẽ cùng khám phá các mẫu giày tây nam Timan cao cấp phù hợp nhất cho môi trường công sở. 1. Giày Derby – Sự Pha Trộn Giữa Cổ Điển Và Hiện Đại Một trong các mẫu giày tây nam được ưa chuộng trong giới công sở là giày Derby. Với thiết kế mở dây, giày Derby mang đến sự thoải mái và dễ dàng trong việc điều chỉnh kích cỡ, phù hợp với những người có mu bàn chân cao. Giày Derby không chỉ thích hợp cho những dịp trang trọng mà còn dễ dàng kết hợp với các trang phục như vest, suit hay thậm chí là quần âu. Đặc điểm của giày Derby nằm ở sự đa dụng và thanh lịch. Bạn có thể chọn giày Derby da trơn cho những ngày làm việc quan trọng hoặc giày Derby da lộn cho phong cách công sở nhẹ nhàng và thoải mái hơn. Màu sắc phổ biến nhất cho giày Derby là đen và nâu, hai gam màu kinh điển mà bất kỳ quý ông nào cũng nên có trong tủ giày. 2. Giày Oxford – Biểu Tượng Của Sự Lịch Lãm Giày Oxford Nam GT80 Giày Oxford luôn nằm trong top đầu các mẫu giày tây nam dành cho môi trường công sở. Với thiết kế dây buộc kín, giày Oxford mang đến vẻ ngoài lịch lãm, sang trọng và rất phù hợp cho các dịp trang trọng như cuộc họp, thuyết trình, hay gặp gỡ đối tác. Điểm khác biệt của giày Oxford so với Derby nằm ở phần dây giày được buộc chặt vào phần thân trên, tạo nên sự gọn gàng và thanh lịch cho đôi chân. Giày Oxford thường được làm từ da cao cấp, tạo nên sự bền bỉ và độ bóng loáng sau thời gian sử dụng. Màu đen là sự lựa chọn phổ biến nhất cho giày Oxford vì tính thanh lịch và dễ kết hợp với trang phục công sở. Ngoài ra, những đôi giày Oxford màu nâu cũng là lựa chọn lý tưởng cho những ai muốn thể hiện phong cách thời trang hiện đại. 3. Giày Loafer – Sự Tiện Lợi Và Thoải Mái Trong số các mẫu giày tây nam cho môi trường công sở, giày Loafer nổi bật với sự tiện lợi và thoải mái. Không cần buộc dây, giày Loafer dễ dàng xỏ vào và tháo ra, giúp tiết kiệm thời gian nhưng vẫn giữ được vẻ ngoài lịch lãm. Giày Loafer được làm từ nhiều chất liệu khác nhau như da bóng, da lộn, hoặc vải cao cấp, mang lại sự đa dạng cho phong cách công sở. Giày Loafer thường được sử dụng trong những môi trường công sở thoải mái hơn, không quá đòi hỏi tính trang trọng. Bạn có thể phối giày Loafer với quần âu, áo sơ mi hoặc cả vest để tạo nên một phong cách công sở đầy phóng khoáng nhưng vẫn chuyên nghiệp. 4. Giày Monk Strap – Phong Cách Sang Trọng Và Độc Đáo Giày Nam Monk Strap GT77 Monk Strap là một trong các mẫu giày tây nam độc đáo, mang lại phong cách riêng biệt cho người mang. Thay vì sử dụng dây buộc truyền thống, giày Monk Strap được thiết kế với khóa cài, tạo nên điểm nhấn sang trọng và khác biệt. Mẫu giày này phù hợp cho những quý ông yêu thích sự phá cách nhưng vẫn muốn giữ được tính trang trọng khi đến văn phòng. Giày Monk Strap có nhiều loại, từ kiểu khóa đơn đến khóa kép. Những đôi giày này thường được làm từ da cao cấp, mang lại độ bền cao và phong cách thời thượng. Màu sắc phổ biến của Monk Strap là đen, nâu và đôi khi là màu burgundy cho những ai muốn tạo nên điểm nhấn trong trang phục. ➔➔➔ Tham khảo những mẫu giày tây nam đẹp, thời thượng được săn đón năm nay: https://plo.vn/nhung-mau-giay-tay-nam-dep-thoi-thuong-duoc-san-don-nam-nay-post754395.html 5. Giày Brogue – Sự Phá Cách Tinh Tế Cuối cùng trong danh sách các mẫu giày tây nam cao cấp cho môi trường công sở là giày Brogue. Đặc điểm nhận diện của giày Brogue chính là những lỗ đục tinh tế trên bề mặt da, tạo nên vẻ ngoài khác biệt và cuốn hút. Giày Brogue thường được lựa chọn bởi những quý ông yêu thích sự sáng tạo trong phong cách, nhưng vẫn muốn giữ sự chuyên nghiệp. Giày Brogue có thể kết hợp với nhiều kiểu trang phục công sở khác nhau, từ quần âu, vest cho đến áo sơ mi. Tuy nhiên, giày Brogue sẽ phù hợp hơn với những dịp công sở không quá trang trọng, nơi bạn có thể thoải mái thể hiện cá tính qua trang phục. Cách Lựa Chọn Giày Tây Nam Cao Cấp Cho Môi Trường Công Sở Giày Oxford Nam GT82 Khi chọn các mẫu giày tây nam cho môi trường công sở, bạn cần lưu ý đến các yếu tố sau để đảm bảo sự phù hợp và thoải mái: Chất liệu: Chất liệu da là lựa chọn hàng đầu khi mua giày tây nam cho công sở. Da bò, da lộn, hoặc da bóng đều mang lại vẻ ngoài sang trọng, bền đẹp theo thời gian. Màu sắc: Màu đen và nâu là hai màu phổ biến nhất cho giày tây nam trong môi trường công sở. Chúng không chỉ dễ phối đồ mà còn tạo nên sự chuyên nghiệp và lịch lãm. Thiết kế: Tùy thuộc vào tính chất công việc, bạn có thể chọn giày buộc dây như Oxford, Derby hoặc giày không dây như Loafer, Monk Strap để phù hợp với phong cách cá nhân và quy định của công ty. Độ thoải mái: Dù mẫu giày có đẹp đến đâu, sự thoải mái vẫn là yếu tố quan trọng nhất. Hãy chọn những đôi giày có độ ôm chân vừa phải, đảm bảo bạn có thể di chuyển cả ngày mà không bị đau chân. Kết Luận Môi trường công sở đòi hỏi sự chuyên nghiệp và chỉn chu trong cách ăn mặc, và giày tây nam là một phần quan trọng trong việc hoàn thiện phong cách của bạn. Dù bạn chọn giày Oxford thanh lịch, Derby thoải mái hay Monk Strap độc đáo, hãy luôn đảm bảo rằng đôi giày không chỉ phù hợp với phong cách cá nhân mà còn mang lại sự thoải mái cho cả ngày dài làm việc. Hy vọng qua bài viết này, bạn đã có thêm nhiều sự lựa chọn trong số các mẫu giày tây nam cao cấp phù hợp cho môi trường công sở, giúp bạn tự tin hơn trong mỗi bước chân.

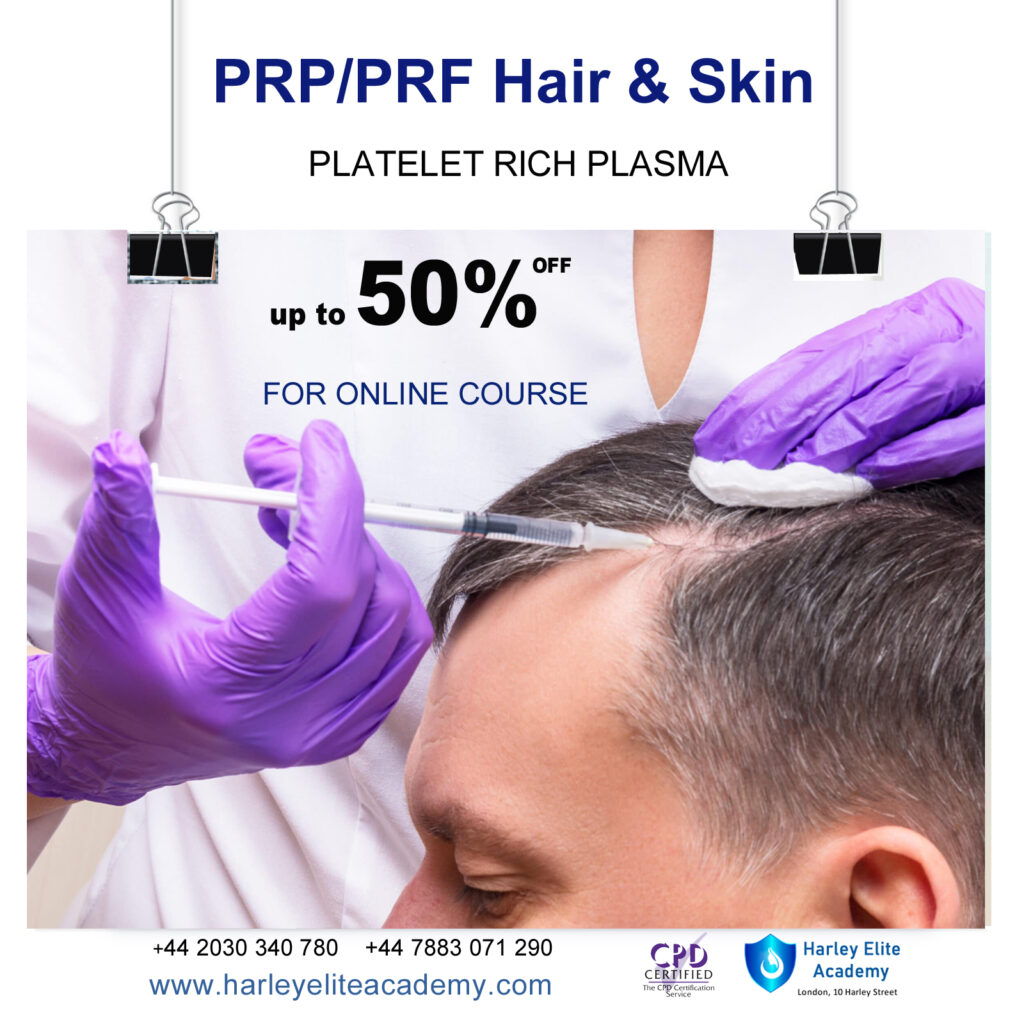
PRP / PRF SKIN & HAIR COURSE
By Harley Elite Academy (HeLa)
MASTERCLASS 8 CPD POINTS 1 DAY INTENSIVE COURSE ONLINE or IN-CLINIC NOTE! After booking we will contact you for scheduling the exact course date! Courses dates are subject to change due to mentors availability. We will inform you via email if a date becomes available! CLINICAL PRP • Sports medicine • Traumatology • Ophthalmic • Burn trauma • Wound healing –diabetic foot • Skin grafting • Dentistry-sinus lift • Tooth implants. PRP theory & equipment: Training Online Theory will enable you to understand: Anatomy Vascular Supply, Contraindications Patient consultation Complications Management Post treatment advice Dealing with equipment A certification of training will be provided upon completion of the course. Aesthetic PRP • Skin rejuvenation • Hair restoration • Fat grafting in combination PRP • Post laser •Acne & Rosacea •Acne scar •TissueVolumisation alternative of HA fillers •Aesthetic gynecology /urology. Plathelet Rich Plasma We will cover pertinent information including mechanism of action, safety and efficacy issues, management and treatment of complications, dilution guidelines, and more. Hands on practical session – skin rejuvenation and hair loss Extraction, Preparation and Dosage Management Injection techniques – face, neck and head (hair loss); also the use of cannula Upon successful completion of the course, you will receive a certificate and title of PRP Certified Practitioner. MASTER CLASS PRP & PRF During the course we are providing . Taking blood and how to use a Centrifuge . PRP injecting techniques in face neck and décolletage hands. PRP Microneedling using a DERMAPEN. Combination treatment PRP with Mesotherapy. MECHANISM OF ACTION Platelets + Leucocytes form 3D mesh release of GF Chemo attraction and migration of macrophages and stem cells Stem cells proliferates by mitosis Stem cells undergo differentiation process BENEFIT FROM PRP TREATMENT & THERAPYExperience the advantages of PRP treatment and therapy, utilizing autologous blood with natural growth factors for disease-free and hypoallergenic benefits. Boost wound healing by regulating mitosis, proliferation, and differentiation, enhancing tissue with collagen, elastin, and hyaluronic acid. Benefit from improved tissue oxygenation, nutrition flow, and support for procedures like hair transplants, fat transfers, and skin grafts.PRP works effectively in skin rejuvenation, facial resurfacing, microneedling, and combines well with HA, PDO threads, skin boosters, peeling, or CO2 lasers. It also proves beneficial for hair restoration, showing positive results in various protocols for Androgenic alopecia and age-related hair loss.PRP where works .Skin rejuvenation-facial resurfacing.application-injection alone. Microneedling Combination with HA,Combination with PDO threads,Skin boosters , peeling or CO2 lasers Hair restoration, Multiple protocols with positive results Evidence for improvement of: Androgenic alopecia-male and females, “spot hair lost” Improvement of age related hair loss. You need to be medically qualified as a doctor, dentist, nurse, pharmacist or paramedic with full governing body registration and have completed a Foundation Filler Course and to have administered a number of cases. Additional information ATTENDANCE ONLINE (Theory), IN CLINIC (Practice) COURSE LEVEL EXPERT | Masterclass Course
Caring for People with Psychosis and Schizophrenia (non-RQF)
By Guardian Angels Training
Gain comprehensive understanding of caring for individuals with psychosis and schizophrenia. Enhance knowledge and skills to provide effective care and support. Designed for caregivers, family members, support workers, and healthcare professionals.

Recognising and Responding to Acutely Unwell Individuals
By Guardian Angels Training
Gain the knowledge and skills to identify acute illness in patients with our "Recognising and Responding to Acutely Unwell Individuals" course. Improve patient outcomes and prevent deterioration in various healthcare settings. Enroll now.

Implementing Good Clinical Laboratory Practice
By Research Quality Association
Course Information Join our comprehensive course, meticulously designed to equip individuals implementing Good Clinical Laboratory Practice (GCLP) within laboratories handling samples from clinical trials. Delve into the current regulatory landscape governing laboratory work supporting clinical trials, referencing key guidelines such as the ICH Guideline for Good Clinical Practice, the Clinical EU Trials Directive, relevant regulations, and leveraging insights from the RQA guidance document on GCLP. Is this course for you? This course is tailored for laboratory managers, analysts, investigators, trial coordinators, monitors, and auditors operating in diverse settings such as pharmaceutical company laboratories, central laboratories, contract research organisations, hospital laboratories, clinics, and investigator sites. This course will give you: Guidance on effectively interpreting and applying GCLP within the broader framework of Good Clinical Practice (GCP) Insight into the seamless integration of GCLP within clinical programmes (GCP) Practical strategies for implementing GCLP in the nuanced environment of clinical research laboratories The chance to update your knowledge with the latest interpretations and guidance on clinical laboratories by the UK Medicines and Healthcare Products Regulatory Agency (MHRA) Access to a seasoned panel of speakers with extensive expertise A unique opportunity to deepen your understanding of GCLP's application across diverse scenarios. Engage in: Lively discussions to foster ideas Problem-solving sessions targeting specific challenges Detailed exploration of specific aspects within the realms of GCP and GCLP. Tutors Tutors will be comprised of (click the photos for biographies): Vanessa Grant -, - Louise Handy Director, Handy Consulting Ltd Tim Stiles Consultant, Qualogy Ltd Programme Please note timings may be subject to alteration. Day 1 08:50 Registration 09:00 Welcome and Introduction 09:20 Good Clinical Practice and the Requirements of Good Clinical Laboratory Practice A review of Good Clinical Practice and its requirements for the laboratory analysis of samples from a trial. The thought processes behind the development of Good Clinical Laboratory Practice, its objective, scope, interpretation and application are explained. 10:00 Safety and Ethical Consideration Informed consent, confidentiality, expedited reporting, blinding and unblinding and serious breaches of the GCP are discussed. 10:40 Break 10:55 Organisation and Personnel Responsibilities within GCP and the Laboratory The responsibilities of key functions that should exist within a clinical laboratory including personnel records of training and competence are discussed. 11:30 Staff Training and Training Records Personnel records of training and competency assessments are discussed. 11:45 Laboratory Facilities, Equipment and Materials Suitable facility design, organisation and operation will be discussed. The calibration, validation and maintenance of equipment used in the conduct of sample analysis are examined, as are the suitability of materials and the identification and labelling of reagents and solutions. 12:30 Lunch 13:15 Workshop 1 - Facilities, Equipment and Responsibilities Some practical problems with regard to the facilities, equipment and responsibilities are explored. 13:45 Workshop 1 - Feedback 14:15 Computer Systems Validation Systems, including computerised systems, used in the analysis, collection and reporting of results should be appropriately tested, operated and controlled. What this means in practice is discussed. 14:45 Trial Protocols, Analytical Plans During this session we examine the purpose, content, control and change of these important documents. 15:30 Break 15:45 Workshop 2 - SOPs, Clinical Protocols, Analytical Plans and Validation The practicalities of managing and documenting the planning phase of analytical work on a trial are explored along with computerised system validation. 16:30 Workshop 2 - Feedback 17:00 Close of Day Day 2 09:00 Conduct of the Work and Quality Control Many of the issues that surround the conduct of sample collection, shipment, storage, analysis and management of Analytical Methods are discussed. This includes the quality control of the assay that may be employed and Quality Control checks. 10:00 Deviation Management The expectations around deviations and CAPA are discussed. 10:15 Workshop 3 - Conduct of the Work and Quality Control Practical work conduct and quality control issues are explored. 10:45 Break 11:00 Workshop 3 - Feedback 11:30 Source Data, Data Integrity, Records and Reports The creation and subsequent management of source data and records, data integrity, are discussed, together with the process of reporting analytical results. 12:10 Workshop 4 - Data, Records and Reports Practical problems with data, records and reports are investigated. 12:45 Lunch 13:30 Workshop 4 - Feedback 14:00 Quality Audit The requirements for and purpose of quality audits are discussed. The difference between quality audit and quality control are explained along with the role of the quality audit staff and their interaction with the analytical project managers, laboratory management and study staff. 14:40 Risk Management How should we assess risk and how can we use the process to assist in evaluation of audit findings. 15:15 Break 15:30 Regulatory Inspection The conduct of regulatory inspections and current expectations of the inspectors. Preparation for inspections and conduct during them will be discussed. 16:00 Panel Session This panel session will address any outstanding issues raised by the delegates. 16:15 Close of Course Extra Information Face-to-face course Course Material Course material will be available in PDF format for delegates attending this course. The advantages of this include: Ability for delegates to keep material on a mobile device Ability to review material at any time pre and post course Environmental benefits – less paper being used per course. The material will be emailed in advance of the course and RQA will not be providing any printed copies of the course notes during the training itself. Delegates wishing to have a hard copy of the notes should print these in advance to bring with them. Alternatively delegates are welcome to bring along their own portable devices to view the material during the training sessions. Remote course Course Material This course will be run completely online. You will receive an email with a link to our online system, which will house your licensed course materials and access to the remote event. Please note this course will run in UK timezone. The advantages of this include: Ability for delegates to keep material on a mobile device Ability to review material at any time pre and post course Environmental benefits – less paper being used per course Access to an online course group to enhance networking. You will need a stable internet connection, a microphone and a webcam. CPD Points 14 Points Development Level Develop

Search By Location
- Pha Courses in London
- Pha Courses in Birmingham
- Pha Courses in Glasgow
- Pha Courses in Liverpool
- Pha Courses in Bristol
- Pha Courses in Manchester
- Pha Courses in Sheffield
- Pha Courses in Leeds
- Pha Courses in Edinburgh
- Pha Courses in Leicester
- Pha Courses in Coventry
- Pha Courses in Bradford
- Pha Courses in Cardiff
- Pha Courses in Belfast
- Pha Courses in Nottingham