- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
8162 Courses
Do you know what to do in case of an accident? Chances are, like most people, you'd also panic and waste a lot of precious time to figure out what needs to be done or what to do while the ambulance arrives. In this paramedicine course, you will learn about paramedicine and paramedicine skills. By the end of this course, you will have a solid understanding of the human anatomy, common injuries- minor and major, infection control and paediatric paramedicine. Furthermore, you will learn the roles and responsibilities of paramedics, how the initial assessment is carried out in case of a medical issue, and how to administer medication to a patient. Having paramedicine skills will ensure you always have valuable skills at your disposal, and you should definitely enrol in this course and add the in-demand skills to your portfolio. This Paramedicine Training Bundle Consists of the following Premium courses: Course 01: Paramedicine Study Course 02: Physiotherapy Assistant Course 03: Cardiac (Heart) Care Course 04: Wound Care Level 2 Course 05: Medical Law Course 06: Medication Administration Level 4 Course 07: GDPR in Healthcare Course 08: Phlebotomy Diploma Course 09: Human Anatomy and Physiology Course 10: Public Health Course 11: Infection Prevention Training Course 12: Personal Hygiene Course 13: Pain Management Course 14: Health and Social Care UK 2021 Enrol now, and start learning Paramedicine skills from the comfort of your home! Curriculum of Paramedicine Bundle Course 01: Paramedicine Study Introduction Roles and Responsibilities of Paramedics in the UK Basic Human Anatomy Initial Assessment Medication Administration Common Injuries Major Injuries Other Medical Emergencies Infection Control Paediatric Paramedicine And many more... How will I get my Paramedicine Certificate? After successfully completing the Paramedicine course you will be able to order your CPD Accredited Certificates (PDF + Hard Copy) as proof of your achievement. PDF Certificate: Free (For The Title Course) Hard Copy Certificate: Free (For The Title Course) CPD 145 CPD hours / points Accredited by CPD Quality Standards Who is this course for? Anyone from any background can enrol in this Paramedicine bundle. Note: Completing this Paramedicine course does not imply that you are qualified to work as a Professional Paramedic. If you are seeking a course that fits the HCPC's standards, please contact the HCPC or the RCSLT ** Requirements Our Paramedicine Training is fully compatible with PC's, Mac's, Laptop, Tablet and Smartphone devices. Career path Having these Paramedicine expertise will increase the value in your CV and open you up to multiple job sectors. Certificates Certificate of completion Digital certificate - Included You will get the PDF Certificate for the title course (Paramedicine Study) absolutely Free! Certificate of completion Hard copy certificate - Included You will get the Hard Copy certificate for the title course (Paramedicine Study) absolutely Free! Other Hard Copy certificates are available for £10 each. Please Note: The delivery charge inside the UK is £3.99, and the international students must pay a £9.99 shipping cost.

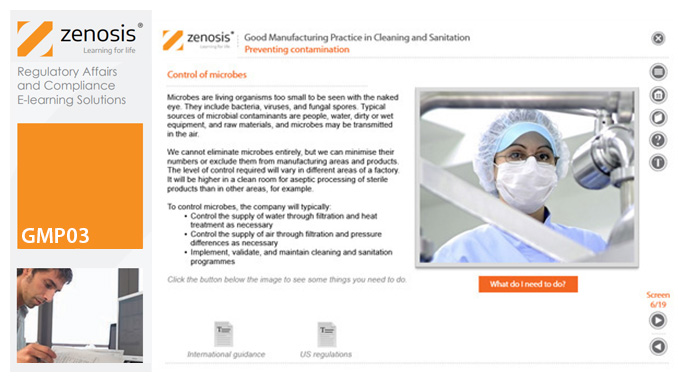
GMP03: Good Manufacturing Practice in Cleaning and Sanitation
By Zenosis
Cleaning and sanitation of premises and equipment are essential to efforts to prevent contamination of product, and they need to be done in compliance with Good Manufacturing Practice (GMP) regulatory requirements. This module shows why it is so important to do a good job, what to consider before and during each job, and how best to go about the work.
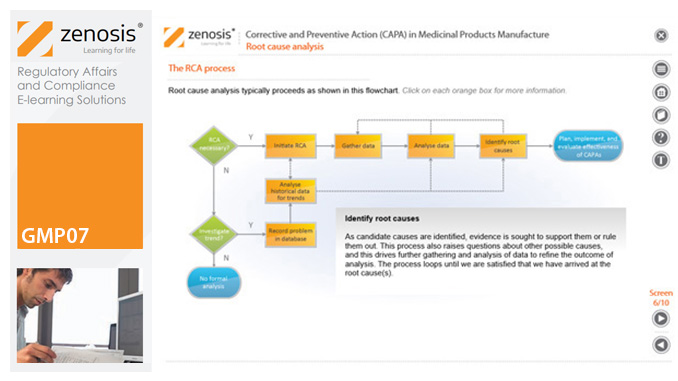
GMP07: Corrective and Preventive Action (CAPA) in Medicinal Products Manufacture
By Zenosis
A company’s Corrective and Preventive Action (CAPA ) system establishes how personnel should deal with manufacturing problems that have occurred or that may occur if not prevented. This module explains the principles of corrective and preventive action and describes typical CAPA procedure. It goes on to introduce root cause analysis and outline the role of progress tracking, escalating, and trending of CAPA procedures.
PV07: Good Pharmacoepidemiology Practice
By Zenosis
Pharmacoepidemiology is the study of the use and effects of drugs in large numbers of people. It provides a bridge between clinical pharmacology and epidemiology. The increasing demand for real-world evidence of the safety, efficacy and utility of medicinal products has focused greater attention on pharmacoepidemiological research. This module will help those who plan and conduct such research, and analyse and report the findings, to follow good practice.

GMP06: Good Manufacturing Practice in Packaging Medicinal Products
By Zenosis
Packaging for medicinal products is subject to Good Manufacturing Practice rules similar to those for the products themselves. In this module we describe the functions that packaging must fulfil and the quality controls that are applied to packaging materials and operations. We set out the requirements for control of printed materials. We describe preparation, in-process control, and completion of a packaging run. Finally, we explain how to carry out reconciliation of packaging materials.

ESS02: Essentials of Monoclonal Antibodies
By Zenosis
This module will introduce you to monoclonal antibodies, explaining how they work, how they are made, and the many uses to which they are put.

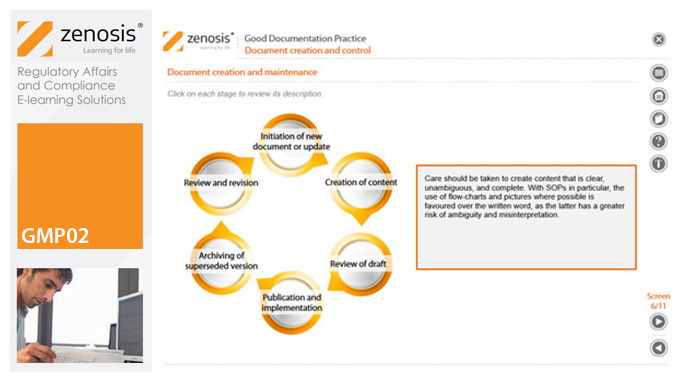
GMP02: Good Documentation Practice
By Zenosis
Good Manufacturing Practice (GMP) for medicinal products relies on documentation. Good Documentation Practice (GDocP) is that part of GMP that applies to the creation, maintenance, use, and retention of documents to provide assurance of the quality of products.
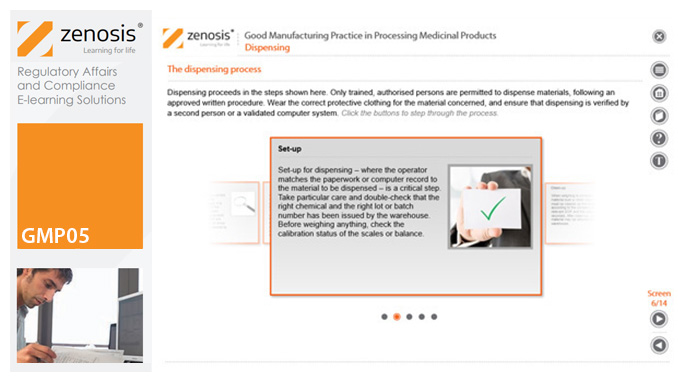
GMP05: Good Manufacturing Practice in Processing Medicinal Products
By Zenosis
Operations in the dispensary and on processing lines are at the heart of medicinal product manufacturing. This module describes how to carry out such operations in compliance with the requirements of Good Manufacturing Practice.
Nursing Assistant Diploma with Adult Nursing - CPD Accredited
By Imperial Academy
Don't wait. The time will never be Just Right

Applied anatomy and physiology Infection control and prevention Different types of tracheostomy Indications for tracheostomy Complications About this event Applied anatomy and physiology Infection control and prevention Different types of tracheostomy Indications for tracheostomy Complications of tracheostomy Changing dressings and keeping the area clean Fitting, managing and changing an inner cannula tube Suctioning a patient with a tracheostomy Communication and care of a patient with a tracheostomy tube Keeping accurate records Full tube change procedure Emergency tracheostomy management (resus) procedure and guidelines Practical session using mannequin & suction machine Training record sign off

Search By Location
- Medicine and Nursing Courses in London
- Medicine and Nursing Courses in Birmingham
- Medicine and Nursing Courses in Glasgow
- Medicine and Nursing Courses in Liverpool
- Medicine and Nursing Courses in Bristol
- Medicine and Nursing Courses in Manchester
- Medicine and Nursing Courses in Sheffield
- Medicine and Nursing Courses in Leeds
- Medicine and Nursing Courses in Edinburgh
- Medicine and Nursing Courses in Leicester
- Medicine and Nursing Courses in Coventry
- Medicine and Nursing Courses in Bradford
- Medicine and Nursing Courses in Cardiff
- Medicine and Nursing Courses in Belfast
- Medicine and Nursing Courses in Nottingham