- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
281 Courses delivered On Demand
CT04f - Clinical study design
By Zenosis
Clinical trial design establishes the framework upon which the clinical trial process will be conducted, and sets the objectives of the trial. The application for marketing approval, submitted to the regulatory authorities, will provide clinical data reflecting the trial design. Since trial design impacts the whole drug development process and lifecycle, particular care and due diligence is essential. This short course provides an overview of the main types of study design.

CT04d - Clinical trial endpoints
By Zenosis
In clinical trials, endpoints are measurements to evaluate the results of a new treatment, at an individual patient level. The study data can be extrapolated to patient populations on the basis of clinical similarities to patients participating in the trial. When clinical trial data have been obtained, focus is on the trial endpoints; more specifically, the focus is on whether the trial met or failed the primary endpoint specified before the trial started. The purpose and various types of endpoints are discussed in this short course.
CT03c - Clinical trial documentation
By Zenosis
Regulatory authorities tend to abide by the maxim that ‘If it isn’t documented, it didn’t happen’. Rigorous documentation of all aspects of a clinical trial is necessary to provide evidence of GCP and compliance with regulatory requirements, as well as enabling effective management of the trial. In this short course we describe important examples of the documents designated by ICH GCP as essential to the conduct of a clinical trial.
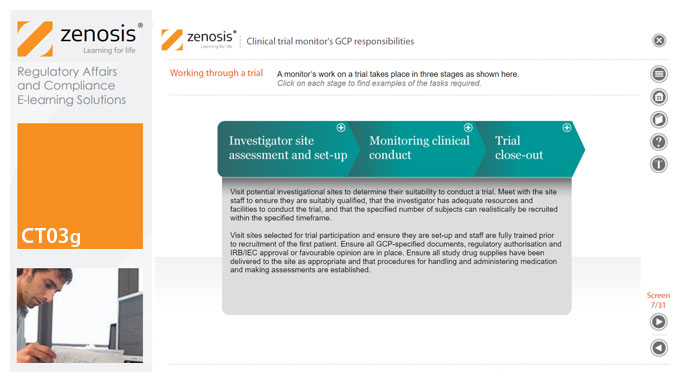
CT03g - Clinical trial monitor’s GCP responsibilities
By Zenosis
A clinical trial monitor acts on behalf of the sponsor to support investigational site personnel, verify the accuracy of data recorded, and ensure that the trial is conducted in compliance with the protocol, GCP and other study specific requirements. He or she acts as the ‘eyes and ears’ of the sponsor at the investigational site and provides the main channel of communication between sponsor and investigator. This short course explores the responsibilities of the monitor and provides insight into key challenges. We discuss assessment of investigators and investigational sites, education and trial initiation, monitoring of clinical conduct, including CRF review and source document verification, and trial close-out. We discuss noncompliance and how to deal with it.
CT04a - Clinical trials in drug development
By Zenosis
New drug development requires major investment in capital, human resources and technical expertise. Strict adherence to regulations on testing and manufacturing standards is also required before a new drug can be marketed. One of the greatest challenges in conducting clinical trials is that of efficiency. As trials become more comprehensive, involving large numbers of participants globally, their duration is prolonged and costs increase. The longer trials last, the shorter is the patent life remaining after market approval and the longer patients must wait for the new product. This short course covers the key components of clinical trials and how these requirements interact with the drug development cycle.

CT03a - ICH, harmonisation, and principles of Good Clinical Practice
By Zenosis
Good Clinical Practice (GCP) is a set of internationally recognised ethical and scientific quality requirements for designing, conducting, recording and reporting clinical trials. Compliance with GCP principles is required by regulatory authorities in many countries for the authorisation of clinical trials and the acceptance of their data in applications for marketing approval. The International Council for Harmonisation's guideline E6, often referred to as ICH GCP, is the international standard specification for Good Clinical Practice. In this short course we describe the ICH’s role in the harmonisation of regulations, introduce its guideline E6, and set out the principles of GCP.
CT04b - Clinical protocol design
By Zenosis
Clinical trial protocols are an essential part of clinical trial design. Protocol documents are critical to conducting safe and cost-effective investigations. Protocol documents are large and complex, containing comprehensive information relating to purpose, design and conduct of a clinical trial. Aspects of a protocol include patient eligibility criteria, and treatment specifications. This short course provides an overview of clinical trial protocols. Opportunities to improve a clinical trial protocol for regulatory approval are also discussed.
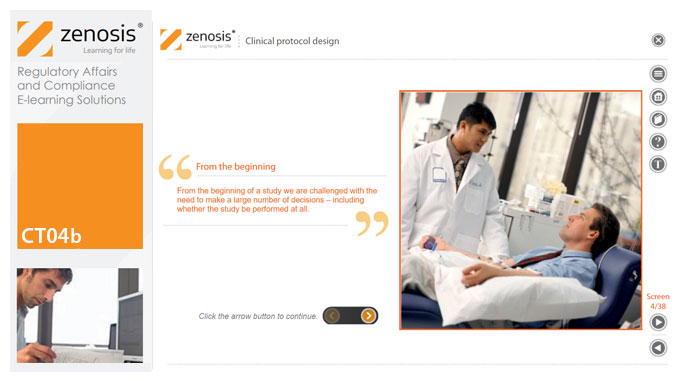
CT03b - Clinical research teamwork
By Zenosis
A clinical trial, particularly a late-phase commercial study, is a major project requiring collaboration between the sponsor and staff or contractor, on the one hand, and the clinical investigator(s) and other healthcare professionals on the other. Good communication among all parties is essential. In this short course we introduce the major roles in a typical clinical research project and outline their duties.
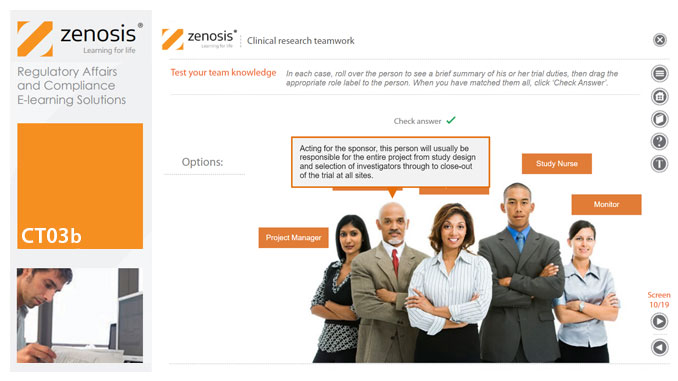
GMP01d - Documentation
By Zenosis
Comprehensive documentation of procedures, formulas, work instructions, and specifications, and thorough recording of batch data, are fundamental requirements of GMP. In this short course we explain why documentation is so important, identify different types of document required, and set out some simple rules for recording and correcting data.
The Midwifery and Maternity Care Training Course offers a thorough exploration of maternal health, guiding learners through the key stages of pregnancy, childbirth, and postnatal support. Whether you're aiming to understand the professional role of midwives or build knowledge relevant to maternity care assistance, this course presents a well-rounded, informative approach. From antenatal care and newborn health to perinatal mental wellbeing and medical complexities during pregnancy, each module is designed to expand your understanding and build your confidence in supporting maternal journeys. This course also covers essential topics such as effective communication, ethical considerations, and reproductive physiology, offering insights into both clinical and compassionate care. You'll examine subjects like twins and higher-order births, contraception, stillbirth support, and terminology used in maternity settings. It’s ideal for those interested in maternal and newborn care, maternity support roles, or simply expanding their healthcare knowledge. Designed for online learning, this training supports flexible study with accessible, structured content built around clarity and relevance. Key Features of Midwifery and Maternity Care Training Bundle CPD Accredited Midwifery and Maternity Care Training Course Instant PDF certificate Fully online, interactive Midwifery and Maternity Care Trainingcourse Self-paced learning and laptop, tablet, smartphone-friendly 24/7 Learning Assistance Discounts on bulk purchases Enrol now in this Midwifery and Maternity Care Training Bundle course to excel! Midwifery and Maternity Care Training Curriculum Midwifery Module 01: Definition and Origin of Midwifery Module 02: Midwifery as a Career Option Module 03: Midwifery Management and Education Module 04: Midwives' Role in Health and Inequality Module 05: Social, Cultural and Spiritual Context of Childbearing Module 06: Antenatal Care and Screening Module 07: Midwifery Support in Labour: Intrapartum Care Module 08: Induction of labour Module 09: Postnatal Care as a Midwife Module 10: Nurture and Nature: The Healthy Newborn Module 11: Stillbirth, Neonatal Death and Bereavement Module 12: Infant feeding Module 13: Newborn Infection Module 14: Home Birth Module 15: Complementary and Alternative Medicines Applied to Maternity Care Module 16: Pharmacology and Medicines Management Module 17: Bleeding in Pregnancy Module 18: Medical Disorders of Pregnancy Module 19: Pre-term Labour Module 20: Twins and higher-order births Module 21: Care During the Third Stage of Labour Module 22: Terminology and Abbreviations for Midwifery Module 23: Perinatal mental health Module 24: Supporting a Healthy Pregnancy Module 25: Contraception and family planning Module 26: Effective Communication for Midwifery Module 27: Ethics in Midwifery Maternity Care Assistant Maternity Care Assistant Module 01: Introduction to Maternity Care Module 02: Terminology and Abbreviations for Maternity Care Assistant Module 03: Reproductive Anatomy & Physiology Module 04: Antenatal Care and Screening Module 05: Support in Labour: Intrapartum Care Module 06: Postnatal Care Module 07: Nurture and Nature: The Healthy Newborn Module 08: Infant feeding Module 09: Newborn Infection Module 10: Care During the Third Stage of Labour Module 11: Perinatal mental health Module 12: Supporting a Healthy Pregnancy Module 13: Effective Communication Module 14: Empathy and Comfort Skills Module 15: Team Working Module 16: Maternity Care Assistance as a Career Accreditation This Midwifery and Maternity Care Training bundle courses are CPD accredited, providing you with up-to-date skills and knowledge and helping you to become more competent and effective in your chosen field. Certification Once you've successfully completed your Midwifery and Maternity Care Training course, you will immediately be sent a digital certificate. Also, you can have your printed certificate delivered by post (shipping cost £3.99). CPD 25 CPD hours / points Accredited by CPD Quality Standards Who is this course for? This course is ideal for all employees or anyone who genuinely wishes to learn more about Midwifery and Maternity Care Training basics. Individuals aspiring to become midwives or maternity care assistants. Healthcare professionals seeking to specialize in maternity care. Students interested in reproductive health and childbirth. Those looking to pursue careers in maternity care facilities. Anyone passionate about supporting healthy pregnancies and newborns. Requirements No prior degree or experience is required to enrol in this Midwifery and Maternity Care Training course. Career path This Midwifery and Maternity Care Training Course will help you to explore avariety of career paths in the related industry. Midwife - £25K to 35K/year. Maternity Support Worker - £18K to 24K/year. Neonatal Nurse - £24K to 40K/year. Obstetrician/Gynaecologist - £40K to 100K/year. Childbirth Educator - £20K to 30K/year Certificates Digital certificate Digital certificate - Included Once you've successfully completed your course, you will immediately be sent a FREE digital certificate. Hardcopy Certificate Hard copy certificate - Included Also, you can have your FREE printed certificate delivered by post (shipping cost £3.99 in the UK). For all international addresses outside of the United Kingdom, the delivery fee for a hardcopy certificate will be only £10. Our certifications have no expiry dates, although we do recommend that you renew them every 12 months.
