- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
200 Credit courses in Cardiff delivered Live Online
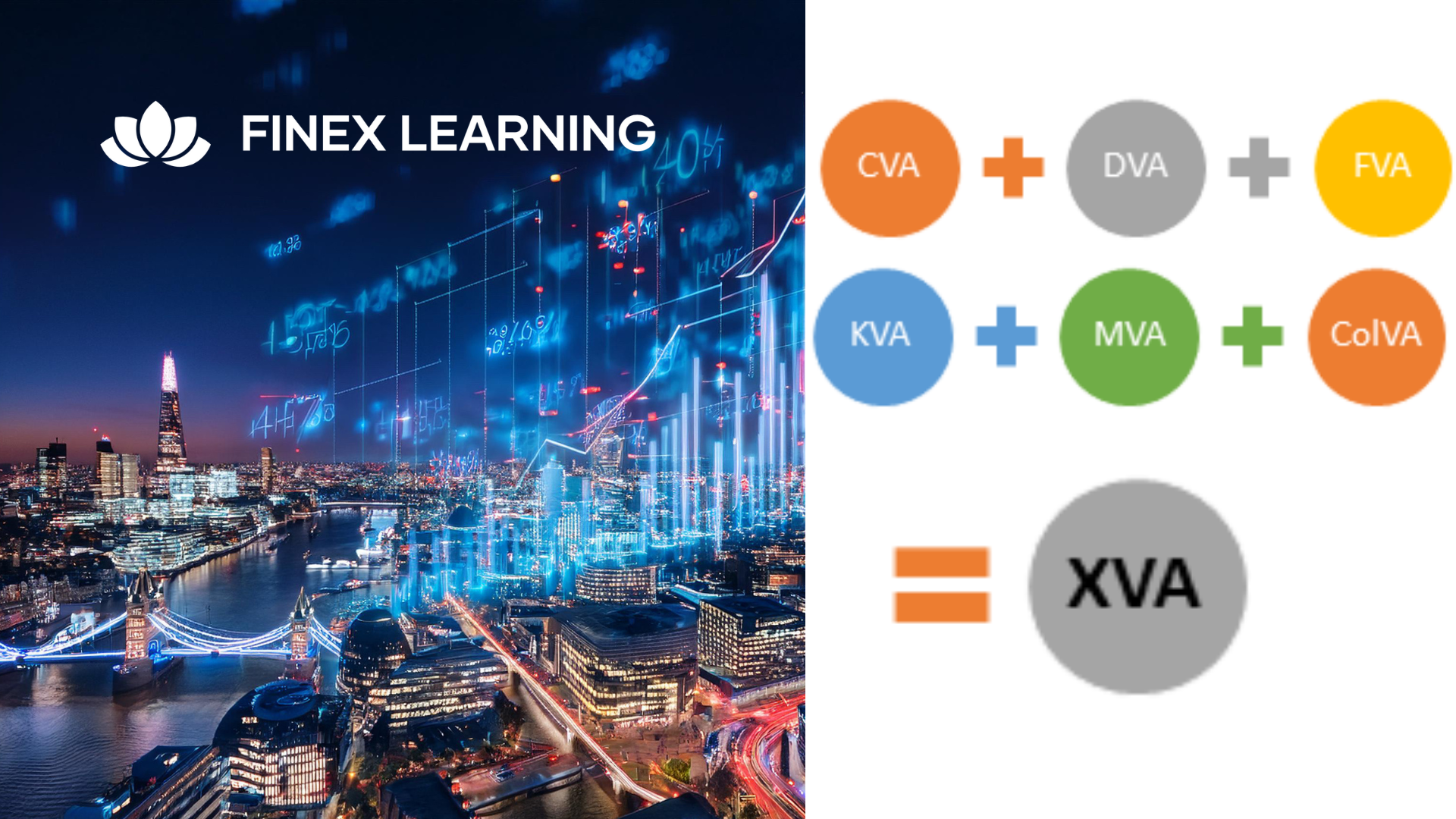
Overview This 1 day course focus on comprehensive review of the current state of the art in quantifying and pricing counterparty credit risk. Learn how to calculate each xVA through real-world, practical examples Understand essential metrics such as Expected Exposure (EE), Potential Future Exposure (PFE), and Expected Positive Exposure (EPE) Explore the ISDA Master Agreement, Credit Support Annexes (CSAs), and collateral management. Gain insights into hedging strategies for CVA. Gain a comprehensive understanding of other valuation adjustments such as Funding Valuation Adjustment (FVA), Capital Valuation Adjustment (KVA), and Margin Valuation Adjustment (MVA). Who the course is for Derivatives traders, structurers and salespeople xVA desks Treasury Regulatory capital and reporting Risk managers (market and credit) IT, product control and legal Quantitative researchers Portfolio managers Operations / Collateral management Consultants, software providers and other third parties Course Content To learn more about the day by day course content please click here To learn more about schedule, pricing & delivery options, book a meeting with a course specialist now
Certificate in Business and Commercial Law (CBCL) - Virtual Learning
By EMG Associates UK Limited
Certificate in Business and Commercial Law (CBCL) - Virtual Learning Why Attend This course is designed for professionals with little or no prior legal background but who are required to make organizational decisions involving legal matters. It will provide participants with the fundamental principles of commercial law, including commercial contracts and negotiation, sale of goods, intellectual property rights and employee relations. It also covers all the legal aspects of setting up a business, running a business and closing a business Participants will gain an in-depth understanding of international commercial law with emphasis on the common law system. Participants will have the opportunity to learn and analyze key legal issues regarding contracts and the business as a whole which they are likely to encounter within their organization. Course Methodology In this interactive training course participants will frequently work in pairs as well as in larger groups to complete exercises as well as regional and international case studies. Course Objectives By the end of the course, participants will be able to: Apply legal rules and principles to speciï¬c commercial situations through consideration of relevant case law Negotiate commercial contracts complying with commercial and legal requirements Increase proï¬tability within their organization by selecting appropriate methods of distribution of goods Recognize and analyze how intellectual property rights aï¬ect their organization Evaluate and modify organizational employment practices complying with labor law requirements Target Audience This course is suitable for those with little or no formal training in commercial law yet would be required to understand the fundamentals of commercial law as it may directly impact their work. It will particularly beneï¬t directors and executives who have direct responsibility for legal decisions within the organization. In-house council new to the region, and those working within a legal department, will also ï¬nd this course highly beneï¬cial. Target Competencies Drafting Contracts Contract Negotiation Understanding Commercial Terms Understanding Employment Law Implementing Organizational Employment Practices Understanding Intellectual Property Understanding corporate restructuring Understanding the effect of breach of commercial contract terms Note The Dubai Government Legal Aï¬airs Department has introduced a Continuing Legal Professional Development (CLPD) programme to legal consultants authorised to practise through a licensed ï¬rm in the Emirate of Dubai. We are proud to announce that the Dubai Government Legal Aï¬airs Department has accredited EMG Associates as a CLPD provider. In addition, all our legal programmes have been approved. This PLUS Specialty Training Legal course qualiï¬es for 4 elective CLPD points. Overview of commercial & business law Areas of commercial and business law Types of commercial contracts Commercial contracts in the civil law and common law systems Sources of English law Civil law v Common law Civil law in the GCC Formalities for a binding contract Elements required for an enforceable contract Rules for contract interpretation implied terms v express terms identifying risks and how to minimise risks Structure of a commercial contract Preliminary documents in international transactions Memorandum of Understanding/ Heads of Terms. Are they legally binding? Commercial implications Boilerplate/ miscellaneous provisions- the important but forgotten clauses- beware! Force majeure v Frustration Notices Set oï¬ No waiver Entire Agreement/ non- reliance clause Times is of the essence Assignment v Novation Governing law Common mistakes in choice of law Jurisdiction clause Exclusive v Non-exclusive Factors in deciding the jurisdiction clause International dispute resolution Litigation Importance of Alternative Dispute Resolution (ADR) Arbitration Mediations Conciliation Negotiation Remedies for breach of contract Damages Specific performance Injunctions Advantages and disadvantages of different business forms Types of business vehicles Sole trader Partnerships Limited Liability Partnerships Companies Cross Border Transactions Distributorship Agency Joint Venture Acquisitions Share purchase v Business purchase Apportioning risks and liabilities through warranties and indemnities Negotiating warranties and indemnities Corporate insolvency Tests identifying insolvency Consequence for directors who fail to react to insolvency Types of insolvency Administration Receivership Creditor Voluntary Liquidation Compulsory Liquidation Commercial Tort Tort of negligence Defamation Libel Slander Recent case law on defamation Managing risk Prevention is better than cure: Eï¬ective risk management Intellectual Property Rights management Types of Intellectual Property copyright trademark patent design rights confidential information Assignment v Granting a license The law of passing off Intellectual Property in the GCC Employment law issues in the common law systems and the GCC Types of employment contracts Grounds for dismissal Wrongful dismissal Redundancy Unfair dismissal Commercial real estate in the GCC Leasehold Freehold

LEVEL 4 ENDORSED CERTIFICATION DIPLOMA
By Export Unlocked Limited
Level 4 Endorsed Diploma - International Trade Qualification Complete all 15 modules and 3 assessments, from a choice of 7 to earn a Level 4 Diploma in International Trade.

Lead Disaster Recovery Manager
By Training Centre
After mastering all the necessary concepts of Disaster Recovery processes, you can sit for the exam and gain the "Certified Lead Disaster Recovery Manager' credential. By holding this Certificate, you will be able to demonstrate that you have the practical knowledge and professional capabilities to support and lead Disaster Recovery teams in implementing Disaster Recovery strategies based on best practices. About This Course Learning objectives Acknowledge the correlation between Disaster Recovery, Business Continuity Management, Information Security and other IT areas and frameworks Master the concepts, approaches, methods and techniques used for the implementation and effective management of a Disaster Recovery Plan Learn how to interpret the ICT Disaster Recovery strategies in the specific context of an organization Learn how to support an organization to effectively plan, implement, manage, monitor and maintain DR services based on best practices Acquire the expertise to advise an organization in implementing an effective Disaster Recovery Plan based on best practices Educational approach This training is based on both theory and best practices used in the implementation and management of a DR plan Lecture sessions are illustrated with examples based on case studies Practical exercises are based on a case study which includes role playing and discussions Practice tests are similar to the Certification Exam Course Content Day 1: Introduction to Disaster Recovery and initiation of a DR plan Day 2: Risk Mitigation Strategies and Disaster Recovery Planning Day 3: Disaster Recovery facilities, services, recovery sites, response and activation Day 4: DRP testing, monitoring, measurement and continuous improvement; the examination Accreditation Prerequisites A foundational understanding of Disaster Recovery Services and knowledge of management principles, concepts and strategies. Assessment The exam for this course consists of 12 essay type questions, to be completed within the 150 minute timeframe and achieving the 70% pass mark. Exam results are provided within 24 hours. What's Included? Certification fees are included on the exam price Training material containing over 450 pages of information and practical examples will be distributed A participation certificate of 31 CPD (Continuing Professional Development) credits will be issued In case of exam failure, you can retake the exam within 12 months for free Provided by This course is Accredited by NACS and Administered by the IECB. Who Should Attend? Information Security Managers Incident Managers Helpdesk Managers Executives with Governance concerns

ISO 13485 Lead Implementer
By Training Centre
During this training course, you will also gain a thorough understanding of the best practices of Medical Devices Quality Management Systems and be able to improve an organization`s overall performance by consistently providing safe and qualitative medical devices. After mastering all the necessary concepts of Medical Devices Quality Management Systems, you can sit for the exam and gain the "Certified ISO 13485 Lead Implementer' Certificate. By holding this Certificate, you will be able to demonstrate that you have the practical knowledge and professional capabilities to implement ISO 13485 in an organization. About This Course Learning Objectives Acknowledge the correlation between ISO 13485 and other standards and regulatory frameworks Master the concepts, approaches, methods and techniques used for the implementation and effective management of a MDQMS Learn how to interpret the ISO 13485 requirements in the specific context of an organization Learn how to support an organization to effectively plan, implement, manage, monitor and maintain a MDQMS Acquire the expertise to advise an organization in implementing Medical Devices Quality Management System best practices Course Agenda Day 1: Introduction to ISO 13485 and initiation of a MDQMS Day 2: Plan the implementation of a MDQMS Day 3: Implementation of a MDQMS Day 4: MDQMS monitoring, measurement, continuous improvement and preparation for a certification audit, and the final exam. Assessment Delegates sit a combined exam, consisting of in-course quizzes and exercises, as well as a final 12 question, essay type exam on Day 4 of the course. The overall passing score is 70%, to be achieved within the 150 minute time allowance. Exam results are provided within 24 hours, with both a Certificate and a digital badge provided as proof of success. Prerequisites A fundamental understanding of ISO 13485 and comprehensive knowledge of implementation principles. What's Included? Certification fees are included on the exam price Training material containing over 450 pages of information and practical examples will be distributed An attestation of course completion worth 32 CPD (Continuing Professional Development) credits will be issued to the participants who have attended the training course. In case of exam failure, you can retake the exam within 12 months for free Who Should Attend? Managers or consultants involved in Medical Devices Quality Management Expert advisors seeking to master the implementation of a Medical Devices Quality Management System Individuals responsible for maintaining conformance with MDQMS requirements MDQMS team members Accreditation Provided by This course is Accredited by NACS and Administered by the IECB

ISO 13485 Lead Auditor
By Training Centre
During this training course, delegates will acquire the knowledge and skills to plan and carry out internal and external audits in compliance with ISO 19011 and ISO/IEC 17021-1 certification process. Based on practical exercises, you will learn audit techniques and become competent to manage an audit program, audit team, communication with customers, and understand reporting requirements. After acquiring the necessary expertise to perform this audit, you can sit for the exam and gain the "Certified ISO 13485 Lead Auditor' Certificate. By holding this Certificate, delegates can demonstrate that they have the competencies to audit organizations based on best practices. About This Course Learning Outcomes Review a Medical Devices Quality Management System based on ISO 13485 Acknowledge the correlation between ISO 13485 and other standards and regulatory frameworks Understand an auditor's role to: plan, lead and follow-up on a management system audit in accordance with ISO 19011 Learn how to lead an audit and audit team Learn how to interpret the requirements of ISO 13485 in the context of a MDQMS audit Acquire the competencies of an auditor to: plan an audit, lead an audit, draft reports, and follow-up on an audit in compliance with ISO 19011 Course Agenda Day 1: Introduction to Medical Devices Quality Management Systems (MDQMS) and ISO 13485 Day 2: Audit principles, preparation and launching of an audit Day 3: On-site audit activities Day 4: Closing the audit and final exam. Assessment Delegates sit a combined exam, consisting of in-course quizzes and exercises, as well as a final essay style exam, consisting of 12 questions, on Day 4 of the course. The overall passing score is 70%, to be achieved within the 150 minute time allowance. Exam results are provided within 24 hours, with both a Certificate and a digital badge provided as proof of success. Prerequisites An understanding of ISO 13485 requirements and knowledge of audit principles. What's Included? Certification fees are included on the exam price Training material containing over 500 pages of information and practical examples will be distributed An attestation of course completion worth 32 CPD (Continuing Professional Development) credits will be issued to the participants who have attended the training course. Who Should Attend? Auditors seeking to perform Medical Devices Quality Management System (MDQMS) certification audits Managers or consultants seeking to understand the Medical Devices Quality Management System audit requirements Individuals responsible for maintaining conformance with Medical Devices Quality Management System requirements Accreditation Provided by This course is Accredited by NACS and Administered by the IECB

ISO 37301 Lead Implementer
By Training Centre
A CMS provides organizations a structured approach to meet all compliance obligations, i.e., requirements that they mandatorily have to comply with such as laws, regulations, court rulings, permits, licenses, as well as those that they voluntarily choose to comply with such as internal policies and procedures, codes of conduct, standards, and agreements with communities or NGOs. About This Course The benefits of implementing a compliance management system (CMS) based on ISO 37301 are manifold: helping the organization avoid or mitigate the costs, risks, and damage of noncompliance, ensuring the long-term sustainability of the organization, promoting trust and confidence, encouraging good governance practices, due diligence, and ethically sound business dealings, etc. The ISO 37301 Lead Implementer training course provides the knowledge needed to establish, implement, manage, maintain, and continually improve a CMS. It aims to provide an in-depth understanding of ISO 37301 requirements, as well as the best practices and approaches used for the implementation and subsequent maintenance of the compliance management system. The training course enables you to help organizations establish processes needed to adhere to all compliance obligations and establish controls that proactively prevent noncompliance and contribute to the creation of a culture of integrity, transparency, and openness. The training course is followed by the certification exam. If you pass, you gain the 'Certified ISO 37301 Lead Implementer' credential. This credential validates your professional capabilities and competences to implement a CMS in an organization based on the requirements of ISO 37301. This training course will help you: Gain a comprehensive understanding of the concepts, approaches, methods, and techniques used for the implementation and effective management of a CMS Acknowledge the correlation between ISO 37301 and other standards and regulatory frameworks Gain the ability to interpret the requirements of ISO 37301 in the specific context of an organization Develop the necessary knowledge and expertise to support an organization in effectively planning, implementing, managing, monitoring, and maintaining a CMS Acquire the expertise to advise an organization in implementing CMS best practices Assessment Delegates sit a combined exam, consisting of in-course quizzes and exercises, as well as a final 12 question, essay type exam on Day 4 of the course. The overall passing score is 70%, to be achieved within the 150 minute time allowance. Exam results are provided within 24 hours, with both a Certificate and a digital badge provided as proof of success. Prerequisites The main requirements for participating in this training course are a basic knowledge of ISO management system standards, as well as a general understanding of ISO 37301 (or ISO 19600 guidelines) and the MS implementation principles. What's Included? Certification fees are included in the exam price. Training material of over 450 pages of information and practical examples will be provided. An attestation of course completion worth 31 CPD (Continuing Professional Development) credits will be issued to participants who have attended the training course. In case of exam failure, candidates can retake the exam once for free within 12 months following the initial exam date. Who Should Attend? Managers, consultants, and compliance officers wishing to develop a thorough understanding of ISO 37301 requirements for a compliance management system Managers and consultants seeking a comprehensive CMS implementation framework Compliance officers responsible for practicing due diligence with regard to compliance risks Individuals wishing to contribute in maintaining organizational integrity by supporting ethical behaviour Managers and members of governance, risk management, and compliance teams Individuals aspiring to become compliance officers or compliance management consultant Accreditation Provided by This course is Accredited by NACS and Administered by the IECB

ISO 37301 Lead Auditor
By Training Centre
Many organizations seek competent auditors to determine whether their policies, procedures, and controls adhere to ISO 37301 requirements. This training course aims to help you complete these tasks successfully and intends to reflect the importance of effective CMS audits. In addition, this training course aims to strengthen your knowledge and skills to plan and carry out CMS audits based on the guidelines for auditing management systems provided in ISO 19011 and the certification process described in ISO/IEC 17021-1. This training course can potentially qualify you to conduct audits on behalf of conformity assessment bodies. The exercises, quizzes, and case studies provided in this training course are designed to help you practice the most important aspects of a CMS audit: ISO 37301 requirements, auditing principles, tools and techniques used to obtain evidence, leading a team of auditors, conducting interviews with auditee, reviewing documented information, drafting nonconformity reports, and preparing the final audit report. After successfully completing the training course, you can sit for the exam and gain the "Certified ISO 37301 Lead Auditor' credential. This credential validates your professional capabilities and demonstrates that you have the knowledge and skills to audit a CMS based on ISO 37301. About This Course This training course enables you to: Understand the main concepts of a compliance management system (CMS) and its processes based on ISO 37301 Acknowledge the relationship between ISO 37301 and other standards and regulatory frameworks Understand the auditor's role in planning, leading, and following up on a CMS audit in accordance with ISO 19011 Interpret the requirements of ISO 37301 in the context of a CMS audit Plan an audit, lead a team of auditors, draft nonconformity reports, and follow up on an audit Act with due professional care during an audit Course Agenda Day 1: Introduction to the compliance management system (CMS) and ISO 37301 Day 2: Audit principles and the preparation for and initiation of an audit Day 3: On-site audit activities Day 4: Closing of the audit and the Exam. Assessment Delegates sit a combined exam, consisting of in-course quizzes and exercises, as well as a final 12 question, essay type exam on Day 4 of the course. The overall passing score is 70%, to be achieved within the 150 minute time allowance. Exam results are provided within 24 hours, with both a Certificate and a digital badge provided as proof of success. Prerequisites A fundamental understanding of ISO 37301 requirements (or ISO 19600 guidelines) for a CMS and a comprehensive knowledge of audit principles. What's Included? Certification fees are included in the exam price. Training material of over 450 pages of information and practical examples. An attestation of course completion worth 31 CPD (Continuing Professional Development) credits will be issued to participants who have attended the training course. In case of exam failure, the candidate can retake the exam once for free within 12 months following the initial exam date. Who Should Attend? The ISO 37301 Lead Auditor training course is intended for: Auditors seeking to perform and lead CMS audits Managers or consultants seeking to master the CMS audit process Individuals responsible for maintaining conformity to ISO 37301 requirements in an organization Technical experts seeking to prepare for a CMS audit Expert advisors and compliance officers Accreditation Provided by This course is Accredited by NACS and Administered by the IECB.

ISO 27035 Lead Incident Manager
By Training Centre
During this training course, you will gain a comprehensive knowledge of a process model for designing and developing an organizational incident management plan. The compatibility of this training course with ISO/IEC 27035 also supports the ISO/IEC 27001 by providing guidance for Information Security Incident Management. After mastering all the necessary concepts of Information Security Incident Management, you can sit for the exam and gain "Certified ISO 27035 Lead Incident Manager" Certification. By holding this certification, you will be able to demonstrate that you have the practical knowledge and professional capabilities to support and lead a team in managing Information Security Incidents. About This Course Learning objectives Master the concepts, approaches, methods, tools and techniques that enable an effective Information Security Incident Management according to ISO/IEC 27035 Acknowledge the correlation between ISO/IEC 27035 and other standards and regulatory frameworks Acquire the expertise to support an organization to effectively implement, manage and maintain an Information Security Incident Response plan Acquire the competence to effectively advise organizations on the best practices of Information Security Incident Management Understand the importance of establishing well-structured procedures and policies for Incident Management processes Develop the expertise to manage an effective Incident Response Team Course Agenda Day 1: Introduction to Information Security Incident Management concepts as recommended by ISO/IEC 27035 Day 2: Designing and preparing an Information Security Incident Management plan Day 3: Enacting the Incident Management process and handling Information Security incidents Day 4: Monitoring and continual improvement of the Information Security Incident Management plan and the Exam. Additional Information Certification fees are included in the exam price. An attendance record worth 31 CPD (Continuing Professional Development) credits will be issued to the participants who have attended the training course. In case candidates fail the exam, they can retake it within 12 months of the initial attempt for free. Accreditation Assessment Delegates sit a combined exam, consisting of in-course quizzes and exercises, as well as a final 12 question, essay type exam on Day 4 of the course. The overall passing score is 70%, to be achieved within the 150 minute time allowance. Exam results are provided within 24 hours, with both a Certificate and a digital badge provided as proof of success. Our Guarantee We are an Accredited Training Provider of the IECB. You can learn wherever and whenever you want with our robust classroom and interactive online training courses. Our courses are taught by qualified practitioners with a minimum of 25 years commercial experience. We strive to give our delegates the hands-on experience. Our courses are all-inclusive with no hidden extras. The one-off cost covers the training, all course materials, and exam voucher. Our aim: To achieve a 100% first time pass rate on all our instructor-led courses. Our Promise: Pass first time or 'train' again for FREE. *FREE training offered for retakes - come back within a year and only pay for the exam. Prerequisites A fundamental understanding of ISO/IEC 27035 and comprehensive knowledge of Information Security. What's Included? Delegates will be provided with; Course Slide deck Questions and Answers Bank Participant Guide Who Should Attend? Information Security Incident managers IT Managers IT Auditors Managers seeking to establish an Incident Response Team (IRT) Managers seeking to learn more about operating effective IRTs Information Security risk managers IT system administration professionals IT network administration professionals Members of Incident Response Teams Individuals responsible for Information Security within an organization Provided by This course is Accredited by NACS and Administered by the IECB

