- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
4856 Courses
November 2025 Fundamentals Organisation & Relationship Systems Coaching Training
By CRR UK
CRRUK equips professionals with the concepts, skills and tools to build conscious, intentional relationships, and to coach relationship systems of any size.

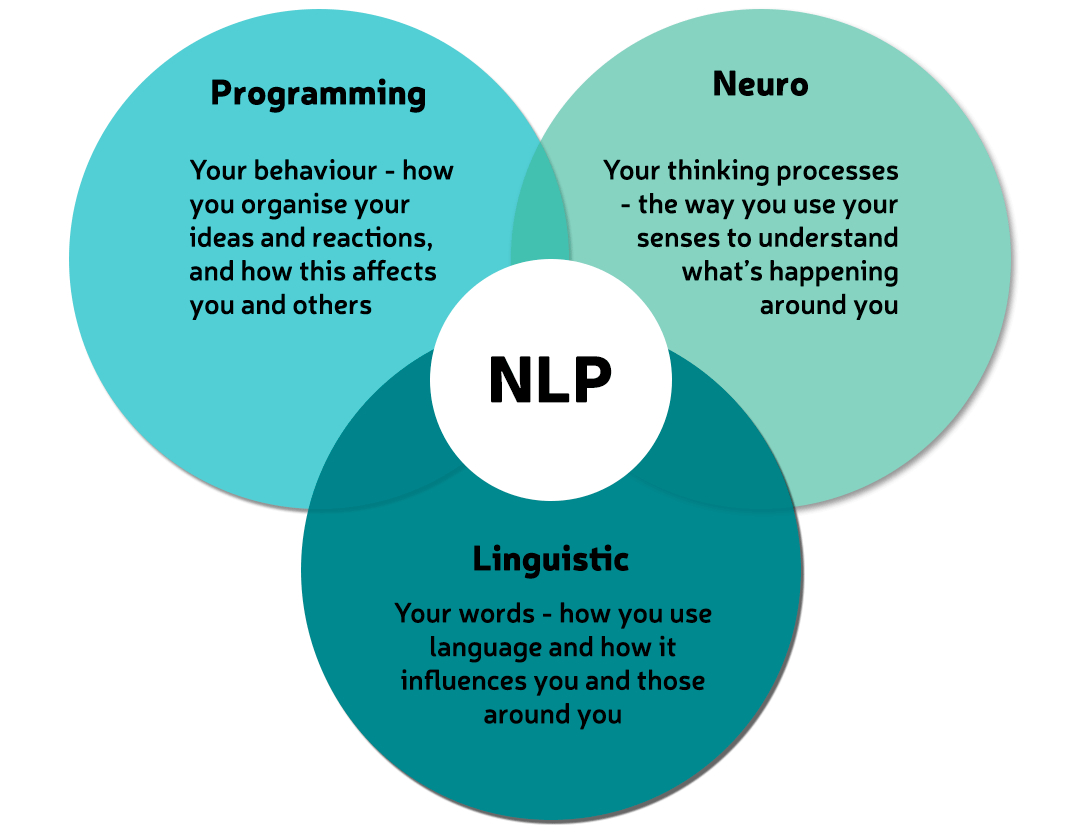
NLP Business Diploma (Fundamentals of Collaborative Working)
By Proactive NLP Ltd
NLP Business Diploma - The Fundamentals of Collaborative Relationships training & certification with Proactive NLP Ltd is your first step towards developing collaborative leadership and collaborative cultures. Start transforming your projects now!
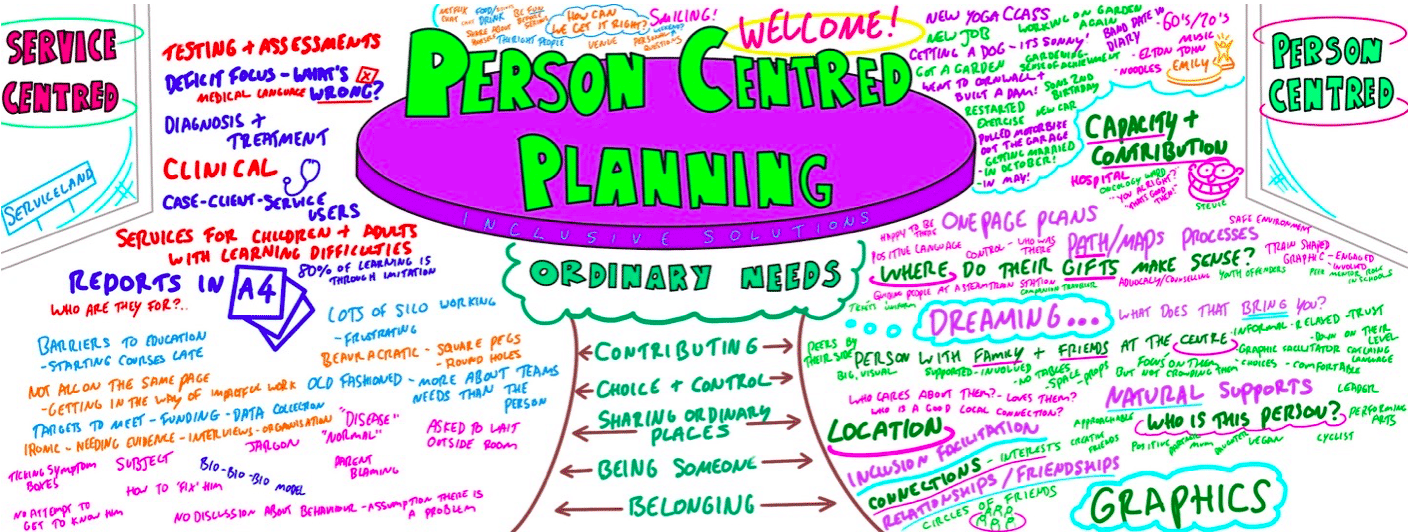
PERSON CENTRED PLANNING TRAINING
By Inclusive Solutions
Person Centred Planning (PCP) is a powerful tool for visioning, future planning and team building. It gets everyone on the same page and creates a progressive, constructive atmosphere so it is especially useful for people who are feeling ‘stuck’ or in a really desperate situation. The PCP processes we teach are called “PATH” and “MAPs”, and are both used for different situations. “To facilitate a group, family, team or organisation in thinking together around a given challenge or issue. Here is an opportunity to experience for real the person centred, futures planning tool – MAP/PATH” (Pearpoint, Forest, O’Brien. 1989). PCP can focus on an individual, with family and friends in the room supporting them, or focus on a group who want to set goals, and realise their potential as a team. PCP focuses on hopes and dreams, so is a very positive approach to planning, and utilises graphic facilitation, making it a very friendly way of working that is accessible to everyone. This unique, hands on PCP training course explores the difference between ‘person-centred thinking’ and ‘service-centred thinking’. Traditionally, we have not always listened deeply enough to the needs of those we are planning for. Often ‘medical model’ thinking dominates our planning for those with the most complex needs. Typically we plan ‘about’ rather than ‘with’ children and adults. We examine this ‘service centred’ approach that most professionals are used to, and question its limitations. This values based approach to inclusion will give participants the ‘theory’ behind why thinking and working in a person centred way is so important, and challenges people to strive for more for the people we are planning around. In addition to the knowledge and theory, this course empowers participants to facilitate their own Person Centred Plans using the PATH or MAPs process and provides the skills to do this effectively. Learning objectives Strengthen understanding of how to think in a person centred way Knowledge of the steps of PATH / MAPs process Ability to facilitate PATH/MAPs process Ability to graphically facilitate a PCP meeting Develop problem solving and planning skills Inclusive Solutions offer: We could provide training for a team of staff, or we could facilitate a PATH or MAP around a disabled person. We can also offer bespoke training packages, please enquire for more details. 1 day “Introduction to PCP” with up to 100 attendees focusing on ‘Person Centred Thinking’ – includes live demonstration of PATH or MAPs Process (for a member of the group / with a student and their family/friends). 2 day “PCP Skills training” – More skills focused, lots of practice. Includes Graphics academy, Process academy and Coaching (Best with under 30 attendees). 2 day “Introduction to PCP with Skills training” – includes live demonstration of PATH or MAPs process, Graphics academy, Process academy and Coaching (Best with under 30 attendees). Half day PATH or MAP for a disabled person, led by two experienced Inclusive Solutions facilitators. Full day PATH or MAP for a team, organisation or group such as a full school staff led by two experienced Inclusive Solutions facilitators. Typical Structure of 2-Day “Introduction to PCP with Skills” Training This course is most useful when delivered over 2 days so we have time to cover the ‘skills’ behind facilitating a PATH or a MAP. Here is an example of what usually happens. If you need us to, we can tailor any of our courses to suit your specific needs. Day 1 AM What makes a good welcome?Service centred thinking and working – what does it involve, how does it make people feel?Reflections on current practices – what is useful and what is harmful?Introduction to underpinning values of person centred thinking and working In depth exploration of the fundamentals of inclusion – encouraging identity, focusing on gifts and capacities Sharing success stories, and personal insight from experienced psychologistsShowcasing a number of practical PCP process in actionIntroduction to the “5 service accomplishments”, or “5 ordinary needs” PM Full PATH process demonstration, led by 2 experienced facilitators – one process facilitator and one graphic facilitatorVolunteers will make notes on what they see the facilitators doing and feedback at the end of the dayFeedback, Q&A Day 2 AM Graphics Academy – we will ease you into the world of graphic facilitation and show you how simple it really is – includes live coaching and graphics tutorial, then participants will practice on each other by facilitating the first 1 or 2 sections of the PATH Process Academy – we will give you some pointers about holding the group, and facilitating in an inclusive way, this is another chance to practice your new graphics skills PM The group is divided up, and volunteers are selected to facilitate PATH meetings The group then run the PATH’s simultaneously with an experienced coaches in the room to guide and to be available for questionsWe reconvene to feedback about the process and digest all we have learntBrief Q&A session and then final reflections
Overview The course begins by introducing credit risk, its components and its drivers. The course then dives into business risk assessment and group structure risks. After examining financial statements, including accounting concepts and principles, the course concludes with a review of financial analysis and its four main risk areas.

Winter Series 2025 - Organisation & Relationship Systems Coaching Training
By CRR UK
CRRUK equips professionals with the concepts, skills and tools to build conscious, intentional relationships, and to coach relationship systems of any size.

Overview This training course will empower you to recognize the root causes of fraud and white-collar crime in the current economy, understand the categories of fraud, equip you with methodologies of fraud detection and prevention, and heighten your ability to detect potential fraudulent situations. In addition to the fundamentals of fraud investigation and detection in a digital environment; profit-loss evaluation, analysis of accounting books, legal concepts, and quantification of financial damages are also examined in this course

It's no secret the world has been changing before our eyes !In order to confront the rapidly-evolving world around us, businesses need highly trained, adaptable and creative individuals to keep pace and stay innovative. BA (Hons) Business Management - 18 months course is designed to train you for the jobs of the future; you'll be taught the fundamentals of business management in the wider context of the contemporary business landscape, ensuring your skills will remain flexible and adaptable as the world changes. This BA (Hons.) Business Management - 18 Months programme aims to develop pro-active decision makers, managers and leaders for a variety of careers in business sectors in a global context. The course combines practical professional experience with creative approaches to enterprise and innovation that will develop your entrepreneurial spirit. Program Overview: BA (Hons) Business Management - 18 MonthsKey Highlights of BA (Hons) Business Management - 18 Months degree programme are: Program Duration: 18 Months (24 Months Option also available) Program Credits: 240 Designed for working Professionals 100% Online Global programme: Study anywhere, anytime on your laptop, phone or tablet. Study material: Comprehensive study materials and e-library support. No Written Exam. The Assessment is done via Submission of Assignment Online Lectures Timely Doubt Resolution Dedicated Student Success Manager Regular Networking Events with Industry Professionals LSBR Alumni Status Payment Plan: No Cost EMI Option

Overview The comprehensive course will cover the fundamentals of portfolio management, and delve deeper into risk and return. Participants will develop their understanding of how and why investors allocate money to fixed income, the numerous issues that impact risk and return, and the mechanics of portfolio construction.

Overview By the end of this Fundamentals of Transfer Pricing Strategies training course, participants will be able to: Acquire basic technical knowledge of transfer pricing Appreciate the importance of the arm's length principle Identify the different stages of a typical transfer pricing analysis Developing a high-level appreciation of the transfer pricing including tax areas and opportunities for multinationals Analyze the current transfer pricing legislative framework Be updated on recent developments in transfer pricing

Overview The course provides fundamentals of business contracts and changes that took place in business contracts. It will train you with the skills to avoid contract drawbacks and understand the documents, agreements or letters of intent and carefully review them before you enter into a contract. It will also train you with the skills required to secure clauses in the contracts to protect your business and to draft effective contracts or even to negotiate amendments with the ultimate aim of protecting your business.

Search By Location
- Fundamentals of Political Science: Core Concepts Courses in London
- Fundamentals of Political Science: Core Concepts Courses in Birmingham
- Fundamentals of Political Science: Core Concepts Courses in Glasgow
- Fundamentals of Political Science: Core Concepts Courses in Liverpool
- Fundamentals of Political Science: Core Concepts Courses in Bristol
- Fundamentals of Political Science: Core Concepts Courses in Manchester
- Fundamentals of Political Science: Core Concepts Courses in Sheffield
- Fundamentals of Political Science: Core Concepts Courses in Leeds
- Fundamentals of Political Science: Core Concepts Courses in Edinburgh
- Fundamentals of Political Science: Core Concepts Courses in Leicester
- Fundamentals of Political Science: Core Concepts Courses in Coventry
- Fundamentals of Political Science: Core Concepts Courses in Bradford
- Fundamentals of Political Science: Core Concepts Courses in Cardiff
- Fundamentals of Political Science: Core Concepts Courses in Belfast
- Fundamentals of Political Science: Core Concepts Courses in Nottingham