- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
2290 Courses
Cognitive Stimulation Therapy (CST) Training online via Zoom
By Dementia Pathfinders Community Interest Company
This one day training will give learners an overview of how to deliver Cognitive Stimulation Therapy (CST). CST is a brief evidence-based group treatment for people with dementia. This interactive course will include a background about CST and how it can be used, research overview and detailed presentation of the treatment, including video footage and role-play.

RCN Accredited Asthma and COPD Course NORFOLK ICB ONLY
By BBO Training
'RCN Accredited Asthma and COPD' CourseThis course is a collaborative offering between your employer and BBO Training Ltd., designed to equip experienced healthcare professionals working in primary care settings with a comprehensive and holistic approach to managing patients with asthma and COPD. Even for those already holding a Diploma in either of these areas, the course has proven to be highly valuable, as indicated by positive evaluations from healthcare professionals.Course AimThe primary objective of this course is to provide qualified healthcare professionals with the necessary knowledge and skills to deliver effective asthma and COPD services in collaboration with their medical colleagues within the community healthcare setting. The course aims to raise awareness of these chronic conditions within the community, ensuring safe practice through the utilization of Patient Group Directions (PGDs) and protocols where appropriate. Participants will be clinically and theoretically prepared to establish and manage review and diagnostic clinics within their competency framework. Furthermore, the course emphasizes the clear differentiation between asthma and COPD.Clinical Practice Program and CompetenciesThroughout the course, you, along with your primary mentor, will organise and validate your clinical engagements, both under supervision and independently. By the time of your final assessment, these forms should be fully completed and ready for submission, accompanied by both your and your mentor's evaluations. Additionally, maintaining a log of all clinical hours and interactions is required, with a minimum of 36 hours.Teaching and Learning ApproachUpon completing this course, students will have the opportunity to:1. Participate in various sessions, including workshops, simulations, discussions, seminars, and tutorials, which can be conducted either virtually or in-person.2. Gain practical experience in asthma and COPD management within their local healthcare settings.3. Develop an in-depth understanding of treatments, including their composition, efficacy, indications, contraindications, and mechanisms of action, through clinical practice.4. Enhance advisory skills related to respiratory care on a broader scale.5. Attend dedicated sessions for clinical practice.6. Document a minimum of 36 hours of supervised clinical practice and maintain an ongoing reflective diary.Expected Course ContentWorkshops, Discussion Groups, Lectures, and Assessment: 40 hoursClinical Practice: A minimum of 36 hoursVisits and Private Study: 44 hoursPrivate study time is essential for reading, conducting factual research on treatments through online sources, and accessing the library for literature searches. This time will facilitate writing, referencing, completing your reflective diary, and preparing for the final assessment.AssessmentSuccessful completion of all components is required. Components 2 to 4 must be passed to complete the course. A resit option is available for any component that is not initially passed, limited to one attempt.1. Reflective Diary: This ongoing document, produced by the student throughout the course, is validated by both the student and mentor. While not directly marked, its completion is integral to your portfolio of evidence.2. Clinical Outcomes: These must be validated and passed by your mentor, with all outcomes achieved, along with a log of clinical hours.3. VIVA: A minimum pass mark of 50% is required.4. Test of Knowledge: A minimum pass mark of 50% is necessary (conducted at the end of the initial study days).Learning OutcomesUpon completion of this course, students will be capable of:1. Evaluating and showcasing clinical competence through a reflective diary/log.2. Demonstrating clinical assessment and treatment proficiency in a VIVA examination and knowledge assessment.3. Effectively educating individuals about self-management and enhancing their understanding of their condition.4. Displaying sensitivity and competence in obtaining comprehensive patient histories.5. Adapting care for diverse patient groups and their unique needs.6. Fulfilling professional responsibilities, including meticulous record-keeping and maintaining confidentiality.7. Teaching the use of various inhalers and assessment equipment.8. Developing fundamental assessment and examination techniques for respiratory conditions.9. Operating with Patient Group Directions and protocols for asthma and COPD care.10. Assessing, planning, implementing, and evaluating individual patient needs comprehensively.
Start learning Clinical Observation Training: Health Safety and Medicine that will give you enough knowledge and skills to build your dream career. About this course This Clinical Observation Training: Health Safety and Medicine helps to grow your skills faster through the power of relevant content and world-class tutors. In this industry-leading bite-sized course, you will learn up-to-date knowledge in the relevant field within a few hours and get certified immediately. The modules of this course are very easy to understand and all of the topics are split into different sections. You will easily grasp and use the knowledge gained from this course in your career and go one step ahead of your competitors. The course is designed to improve your employability and provide you with the tools you need to succeed. Enrol today and start learning your essential skills. Why choose this course Earn a digital Certificate upon successful completion. Accessible, informative modules taught by expert instructors Study in your own time, at your own pace, through your computer tablet or mobile device Get 24/7 help or advice from our email and live chat teams Get full tutor support on weekdays (Monday to Friday) Course Design The Clinical Observation Training: Health Safety and Medicine is delivered through our online learning platform, accessible through any internet-connected device. There are no formal deadlines or teaching schedules, meaning you are free to study the course at your own pace. You are taught through a combination of Video lessons Online study materials Who Is This Course For:â This Clinical Observation Training: Health Safety and Medicine is ideal for those who want to be skilled in this field or who wish to learn a new skill to build their dream career. If you want to gain extensive knowledge, potential experience, and be an expert in the related field then this is a great course for you to grow your career. Requirements This course is for anyone who would like to learn Clinical Observation Training: Health Safety and Medicine related skills to aid his/her career path. No formal entry prerequisites are required Certification Upon successful completion of the course, you will be able to obtain your course completion e-certificate. Print copy by post is also available at an additional cost of £9.99 and PDF Certificate at £4.99. Course Content Module 01: An Overview of Clinical Observation and Clinical Career Module 02: Observation of Clinical Management Module 03: Observation of technical clinical method Module 04: Observation of Medicine and Pharmacies Module 05: Hygiene and Health Safety Course Content Clinical Observation: Health Safety and Medicine Module 01: An Overview of Clinical Observation and Clinical Career 00:08:00 Module 02: Observation of Clinical Management 00:13:00 Module 03: Observation of technical clinical method 00:20:00 Module 04: Observation of Medicine and Pharmacies 00:10:00 Module 05: Hygiene and Health Safety 00:17:00 Order your Certificates & Transcripts Order your Certificates & Transcripts 00:00:00 Frequently Asked Questions Are there any prerequisites for taking the course? There are no specific prerequisites for this course, nor are there any formal entry requirements. All you need is an internet connection, a good understanding of English and a passion for learning for this course. Can I access the course at any time, or is there a set schedule? You have the flexibility to access the course at any time that suits your schedule. Our courses are self-paced, allowing you to study at your own pace and convenience. How long will I have access to the course? For this course, you will have access to the course materials for 1 year only. This means you can review the content as often as you like within the year, even after you've completed the course. However, if you buy Lifetime Access for the course, you will be able to access the course for a lifetime. Is there a certificate of completion provided after completing the course? Yes, upon successfully completing the course, you will receive a certificate of completion. This certificate can be a valuable addition to your professional portfolio and can be shared on your various social networks. Can I switch courses or get a refund if I'm not satisfied with the course? We want you to have a positive learning experience. If you're not satisfied with the course, you can request a course transfer or refund within 14 days of the initial purchase. How do I track my progress in the course? Our platform provides tracking tools and progress indicators for each course. You can monitor your progress, completed lessons, and assessments through your learner dashboard for the course. What if I have technical issues or difficulties with the course? If you encounter technical issues or content-related difficulties with the course, our support team is available to assist you. You can reach out to them for prompt resolution.
CT04a - Clinical trials in drug development
By Zenosis
New drug development requires major investment in capital, human resources and technical expertise. Strict adherence to regulations on testing and manufacturing standards is also required before a new drug can be marketed. One of the greatest challenges in conducting clinical trials is that of efficiency. As trials become more comprehensive, involving large numbers of participants globally, their duration is prolonged and costs increase. The longer trials last, the shorter is the patent life remaining after market approval and the longer patients must wait for the new product. This short course covers the key components of clinical trials and how these requirements interact with the drug development cycle.

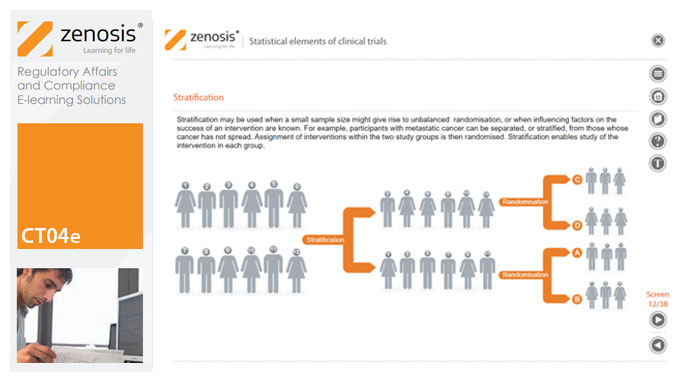
CT04e - Statistical elements of clinical trials
By Zenosis
Analytical statistical elements are essential concepts in the design of clinical trials. This analysis helps us to understand whether a conclusion from a study of a sample of the target population applies generally to that population as a whole. In particular, it helps us to answer the question: Did the treatment effect in the given study occur just by chance? The statistical elements of a well-controlled study minimise the chances of drawing the wrong conclusions, by providing clear thresholds for such errors. The basic statistical elements of a clinical trial include eligibility criteria, randomisation, sample size, power, and blinding, and these are discussed in this short course.
CT13: Safety Reporting in Clinical Trials
By Zenosis
This course explains the regulatory requirements for the reporting of adverse events and suspected adverse reactions in clinical trials. It describes how investigators should report to sponsors, and how sponsors should report to regulatory authorities and other stakeholders in the safety of investigational products. It explains how events are characterized as serious or non-serious, expected or unexpected, and it distinguishes the requirements for each category. It describes controlled vocabularies used for coding of events in reports.
Laboratory Technician Level 7 - QLS Endorsed
By Kingston Open College
QLS Endorsed + CPD QS Accredited - Dual Certification | Instant Access | 24/7 Tutor Support | All-Inclusive Cost

Elevate your career in healthcare with our comprehensive Clinical Chemistry and Medical Laboratory Technician course. Gain expertise in laboratory techniques, clinical chemistry analysis, immunology, and more. Ensure precision with quality assurance, laboratory safety, and cutting-edge technologies. Join us for hands-on training and advance your skills in a dynamic and vital field. Enroll now for a rewarding journey in medical laboratory services.

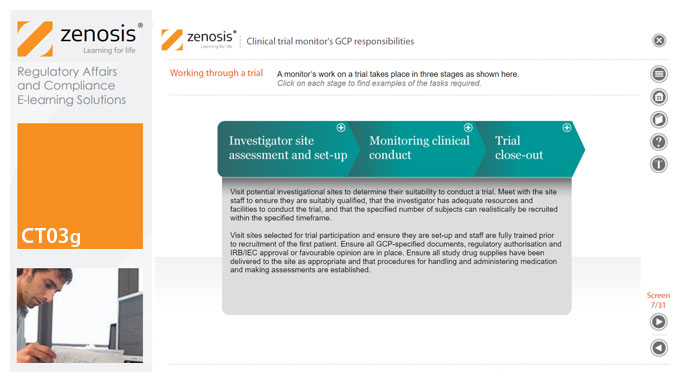
CT03g - Clinical trial monitor’s GCP responsibilities
By Zenosis
A clinical trial monitor acts on behalf of the sponsor to support investigational site personnel, verify the accuracy of data recorded, and ensure that the trial is conducted in compliance with the protocol, GCP and other study specific requirements. He or she acts as the ‘eyes and ears’ of the sponsor at the investigational site and provides the main channel of communication between sponsor and investigator. This short course explores the responsibilities of the monitor and provides insight into key challenges. We discuss assessment of investigators and investigational sites, education and trial initiation, monitoring of clinical conduct, including CRF review and source document verification, and trial close-out. We discuss noncompliance and how to deal with it.
Search By Location
- Clinical Courses in London
- Clinical Courses in Birmingham
- Clinical Courses in Glasgow
- Clinical Courses in Liverpool
- Clinical Courses in Bristol
- Clinical Courses in Manchester
- Clinical Courses in Sheffield
- Clinical Courses in Leeds
- Clinical Courses in Edinburgh
- Clinical Courses in Leicester
- Clinical Courses in Coventry
- Clinical Courses in Bradford
- Clinical Courses in Cardiff
- Clinical Courses in Belfast
- Clinical Courses in Nottingham
