- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
294 Courses delivered Online
Retrain ONLINE for a Higher-Paying Job in the Pharma Manufacturing Industry in 10-Weeks
By Getreskilled (UK)
Average entry-level pharma roles pay 96% of NHS Band 5 and come with opportunities for overtime, shift allowance and additional benefits as well as huge scope for career progression and salary increases.

Layer of Protection Analysis (LOPA)
By EnergyEdge - Training for a Sustainable Energy Future
Take your knowledge to the next level with EnergyEdge course on Layer of Protection Analysis (LOPA). Enroll now to advance your career.

24 Hour Flash Deal **25-in-1 Pharmaceutical Sales Representative Diploma Mega Bundle** Pharmaceutical Sales Representative Diploma Enrolment Gifts **FREE PDF Certificate**FREE PDF Transcript ** FREE Exam** FREE Student ID ** Lifetime Access **FREE Enrolment Letter ** Take the initial steps toward a successful long-term career by studying the Pharmaceutical Sales Representative Diploma package online with Studyhub through our online learning platform. The Pharmaceutical Sales Representative Diploma bundle can help you improve your CV, wow potential employers, and differentiate yourself from the mass. This Pharmaceutical Sales Representative Diploma course provides complete 360-degree training on Pharmaceutical Sales Representative Diploma. You'll get not one, not two, not three, but twenty-five Pharmaceutical Sales Representative Diploma courses included in this course. Plus Studyhub's signature Forever Access is given as always, meaning these Pharmaceutical Sales Representative Diploma courses are yours for as long as you want them once you enrol in this course This Pharmaceutical Sales Representative Diploma Bundle consists the following career oriented courses: Course 01: Medical Sales Representative Course 02: Pharmaceutical Marketing, Product Development & and Medical Engagement Course 03: Pharmacology Diploma Course 04: Sales Skills Course 05: Advanced Sales and Selling Management Techniques Course 06: Key Account Management (KAM) Certificate Course 07: Medical Terminology Training Course 08: Business Development Manager Course 09: Customer Relationship Management Course 10: Sales Analysis in Excel Course 11: Negotiation Skills Course 12: Product Management for Maximum Sales: Boosting Business Success Course 13: Medical Receptionist Course 14: Human Anatomy, Physiology and Medical Terminology Diploma Course 15: Financial Planning Course 16: Professional Digital Marketing Diploma Course 17: Medical Law Course 18: Healthcare GDPR Certificate Course 19: Essentials of European Medical Device Regulations Course 20: Health Economics and Health Technology Assessment Course 21: International Healthcare Policy Course 22: Social Media Marketing Course Course 23: Marketing Strategies for Business Course 24: Effective Communication Skills Diploma Course 25: Business Networking Skills The Pharmaceutical Sales Representative Diploma course has been prepared by focusing largely on Pharmaceutical Sales Representative Diploma career readiness. It has been designed by our Pharmaceutical Sales Representative Diploma specialists in a manner that you will be likely to find yourself head and shoulders above the others. For better learning, one to one assistance will also be provided if it's required by any learners. The Pharmaceutical Sales Representative Diploma Bundle is one of the most prestigious training offered at StudyHub and is highly valued by employers for good reason. This Pharmaceutical Sales Representative Diploma bundle course has been created with twenty-five premium courses to provide our learners with the best learning experience possible to increase their understanding of their chosen field. This Pharmaceutical Sales Representative Diploma Course, like every one of Study Hub's courses, is meticulously developed and well researched. Every one of the topics is divided into Pharmaceutical Sales Representative Diploma Elementary modules, allowing our students to grasp each lesson quickly. The Pharmaceutical Sales Representative Diploma course is self-paced and can be taken from the comfort of your home, office, or on the go! With our Student ID card you will get discounts on things like music, food, travel and clothes etc. In this exclusive Pharmaceutical Sales Representative Diploma bundle, you really hit the jackpot. Here's what you get: Step by step Pharmaceutical Sales Representative Diploma lessons One to one assistance from Pharmaceutical Sales Representative Diplomaprofessionals if you need it Innovative exams to test your knowledge after the Pharmaceutical Sales Representative Diplomacourse 24/7 customer support should you encounter any hiccups Top-class learning portal Unlimited lifetime access to all twenty-five Pharmaceutical Sales Representative Diploma courses Digital Certificate, Transcript and student ID are all included in the price PDF certificate immediately after passing Original copies of your Pharmaceutical Sales Representative Diploma certificate and transcript on the next working day Easily learn the Pharmaceutical Sales Representative Diploma skills and knowledge you want from the comfort of your home CPD 250 CPD hours / points Accredited by CPD Quality Standards Who is this course for? This Pharmaceutical Sales Representative Diploma training is suitable for - Students Recent graduates Job Seekers Individuals who are already employed in the relevant sectors and wish to enhance their knowledge and expertise in Pharmaceutical Sales Representative Diploma Requirements To participate in this Pharmaceutical Sales Representative Diploma course, all you need is - A smart device A secure internet connection And a keen interest in Pharmaceutical Sales Representative Diploma Career path You will be able to kickstart your Pharmaceutical Sales Representative Diploma career because this course includes various courses as a bonus. This Pharmaceutical Sales Representative Diploma is an excellent opportunity for you to learn multiple skills from the convenience of your own home and explore Pharmaceutical Sales Representative Diploma career opportunities. Certificates CPD Accredited Certificate Digital certificate - Included CPD Accredited e-Certificate - Free CPD Accredited Hardcopy Certificate - Free Enrolment Letter - Free Student ID Card - Free

24 Hour Flash Deal **25-in-1 Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences Mega Bundle** Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences Enrolment Gifts **FREE PDF Certificate**FREE PDF Transcript ** FREE Exam** FREE Student ID ** Lifetime Access **FREE Enrolment Letter ** Take the initial steps toward a successful long-term career by studying the Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences package online with Studyhub through our online learning platform. The Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences bundle can help you improve your CV, wow potential employers, and differentiate yourself from the mass. This Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences course provides complete 360-degree training on Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences. You'll get not one, not two, not three, but twenty-five Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences courses included in this course. Plus Studyhub's signature Forever Access is given as always, meaning these Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences courses are yours for as long as you want them once you enrol in this course This Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences Bundle consists the following career oriented courses: Course 01: Pharmacy Assistant & Pharmacy Technician Course 02: Diploma in Pharmacy Skills Course 03: Medication Administration: Control and Administration of Medicine Course 04: Medication Administration and Preservation Course 05: Medical Transcription Diploma Course 06: Nurse Prescribing and Medicine Management Course 07: Immunisation Nurse Course 08: Pharmacology Diploma Course 09: Healthcare GDPR Certificate Course 10: Biomedical Science Course 11: Medical Secretary Certification Course 12: Diploma in Laboratory Technician Course 13: Medical Receptionist Course 14: Medical & Clinical Administrator Course 15: Medical Law Course 16: Essentials of European Medical Device Regulations Course 17: Medical Writing Course 18: Phlebotomy Course 19: Observation Skills for Carers Course 20: Pain Management Course 21: English for Healthcare Course 22: Common Childhood Illnesses & Paediatric First Aid Course 23: Diabetes Awareness Training Course 24: Clinical Research Administration: Navigating the Healthcare Landscape Course 25: Personal Hygiene The Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences course has been prepared by focusing largely on Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences career readiness. It has been designed by our Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences specialists in a manner that you will be likely to find yourself head and shoulders above the others. For better learning, one to one assistance will also be provided if it's required by any learners. The Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences Bundle is one of the most prestigious training offered at StudyHub and is highly valued by employers for good reason. This Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences bundle course has been created with twenty-five premium courses to provide our learners with the best learning experience possible to increase their understanding of their chosen field. This Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences Course, like every one of Study Hub's courses, is meticulously developed and well researched. Every one of the topics is divided into Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences Elementary modules, allowing our students to grasp each lesson quickly. The Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences course is self-paced and can be taken from the comfort of your home, office, or on the go! With our Student ID card you will get discounts on things like music, food, travel and clothes etc. In this exclusive Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences bundle, you really hit the jackpot. Here's what you get: Step by step Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences lessons One to one assistance from Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciencesprofessionals if you need it Innovative exams to test your knowledge after the Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciencescourse 24/7 customer support should you encounter any hiccups Top-class learning portal Unlimited lifetime access to all twenty-five Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences courses Digital Certificate, Transcript and student ID are all included in the price PDF certificate immediately after passing Original copies of your Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences certificate and transcript on the next working day Easily learn the Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences skills and knowledge you want from the comfort of your home CPD 250 CPD hours / points Accredited by CPD Quality Standards Who is this course for? This Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences training is suitable for - Students Recent graduates Job Seekers Individuals who are already employed in the relevant sectors and wish to enhance their knowledge and expertise in Pharmacy technician Training: Advanced Skills in Pharmaceutical Sciences Requirements To participate in this Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences course, all you need is - A smart device A secure internet connection And a keen interest in Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences Career path You will be able to kickstart your Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences career because this course includes various courses as a bonus. This bundle is an excellent opportunity for you to learn multiple skills from the convenience of your own home and explore Pharmacy Technician Training: Advanced Skills in Pharmaceutical Sciences career opportunities. Certificates CPD Accredited Certificate Digital certificate - Included CPD Accredited e-Certificate - Free CPD Accredited Hardcopy Certificate - Free Enrolment Letter - Free Student ID Card - Free

Introduction to Good Manufacturing Practice
By Research Quality Association
Course Information This course offers foundational guidance and practical support tailored for individuals operating within Good Manufacturing Practice (GMP) frameworks. Explore the fundamental prerequisites of a pharmaceutical quality system (PQS) and delve into the application of quality risk management (QRM) principles, aligning with current regulations and guidance. Gain insights into pivotal aspects such as requirements, roles, and responsibilities, encompassing change control, document management, and key documentation essential for effective implementation of GMP with a focus on regulatory inspections and common findings. Is this course for you? Ideal for professionals engaged in GMP across various sectors, including: Research and Development (R&D) Contract Manufacturing Organisations Manufacturing Units Quality Control (QC) Laboratories Auditing Roles. What will you learn? Event objectives - by the end of the course, delegates shall: Have an awareness of the basic requirements of GMP Be aware of UK and EU GMP Rules and Guidance and relevant publications Understand the roles and responsibilities associated with GMP Be able to contribute to and maintain quality documentation Have a basic understanding of product lifecycle and manufacturing Understand the requirements of GMP in the QC laboratory context Have a basic understanding of risk management and mitigation principles Understand the need for quality systems and quality assurance activities Be aware of common regulatory findings. Learning outcomes: delegates will be able to: Implement their role within GMP with confidence and knowledge of the principle requirements Contribute effectively to the GMP quality system and their organisation’s compliance Comprehend where their organisation’s activities sit within the larger GMP arena Know where to seek further information within the published rules and guidance, UK Legislation, European Commission Directives, ICH Guidance and other relevant publications, as well as via the internet. Tutors Tutors will be comprised of (click the photos for biographies): Louise Handy Director, Handy Consulting Ltd Programme Please note timings may be subject to alteration. Day 1 09:30 Introductions and Scope of the Course Understand the group requirements and the tutor's background and experience. 09:45 Background and Regulatory Environment Setting the scene, understanding the context, key legislation. 10:30 Principles of GMP Key points and requirements. 11:15 Break 11:30 Personnel and Responsibilities Management and staff, duties and accountabilities. 12:00 Overview of GMP Manufacturing Basics of the product life cycle. 12:30 Lunch 13:15 Risk Management Workshop Practical exploration of risk and mitigation activities. 14:30 QC Laboratories Activities and practicalities. 15:15 Break 15:30 Compliance Quality Assurance and Self Inspection. 16:15 Question Time A chance for questions on the practicalities of GMP. 16:30 Close of Course Extra Information Course Material This course will be run completely online. You will receive an email with a link to our online system, which will house your licensed course materials and access to the remote event. Please note this course will run in UK timezone. The advantages of this include: Ability for delegates to keep material on a mobile device Ability to review material at any time pre and post course Environmental benefits – less paper being used per course Access to an online course group to enhance networking. You will need a stable internet connection, a microphone and a webcam. CPD Points 7 Points Development Level Learn

PV03: Drug Safety and Pharmacovigilance
By Zenosis
Drug safety monitoring and risk management are vitally important for medicinal product developers, licence holders and clinical investigators. In addition to their duty to protect public health, increasingly tight regulation and potentially massive payments to litigants provide strong incentives for pharmaceutical and biotechnology companies to ensure that they maintain efficient systems for drug safety / pharmacovigilance and that all staff are aware of the basic requirements. This course will provide them with an overview of the most important aspects of this discipline, both before and after marketing of products, especially as they apply in Europe and the USA.

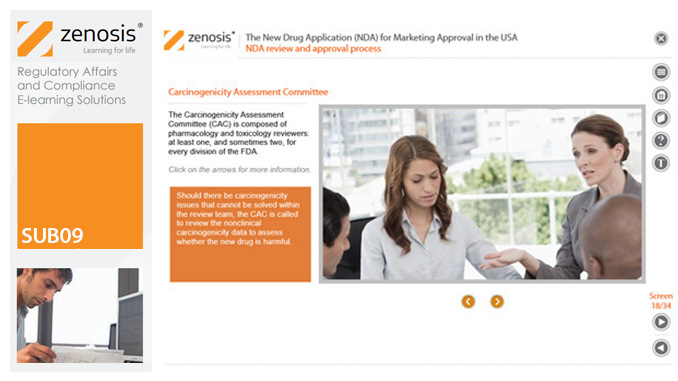
SUB09: The New Drug Application (NDA) for Marketing Approval in the USA
By Zenosis
The New Drug Application (NDA) is the regulatory vehicle through which sponsors formally propose that the Food and Drug Administration (FDA) approve a new pharmaceutical for marketing and sale in the USA.
VAL07: Computer Systems Validation, Part 2: Implementation
By Zenosis
This module describes the design, development and installation phase, the validation phase, and the operation and maintenance phase of the validation of computerised systems in medicines and healthcare products manufacturing environments. It continues to follow the progress of a pharmaceutical company's project to validate a new dispensary control system.
VAL06: Computer Systems Validation, Part 1: Planning
By Zenosis
In the medicines and healthcare products industries, computerised systems used in automated manufacturing or laboratory processes to which Good Manufacturing Practice requirements apply need to be validated. This module describes the planning of such validation. It follows the work of a pharmaceutical company's team as they validate the dispensary control system for a new production line.

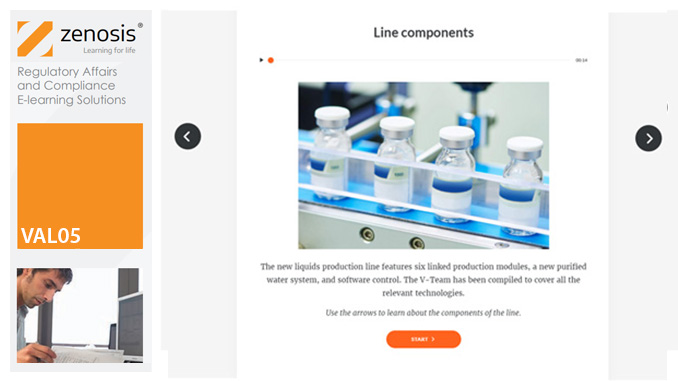
VAL05: Equipment Cleaning Validation
By Zenosis
Manufacturers of medicines and healthcare products must establish, validate and maintain an equipment cleaning programme. This is a regulatory requirement because validated cleaning procedures contribute to the assurance of product purity and safety. This module provides a comprehensive account of equipment cleaning validation requirements and procedures. It follows the work of a pharmaceutical company's validation team as they establish and validate the cleaning program for a new production line.