- Professional Development
- Medicine & Nursing
- Arts & Crafts
- Health & Wellbeing
- Personal Development
Certified Scrum Professional-ScrumMaster (CSP-SM)
By IIL Europe Ltd
Certified Scrum Professional®-ScrumMaster® (CSP®-SM) Certified Scrum Professionals challenge their teams to improve the way Scrum and Agile principles are applied. They have demonstrated experience, documented training, and proven knowledge in Scrum. Are you ready to take your knowledge and skillset in your role as Scrum Master to the next level? If so, it's time to elevate your career further by earning the Certified Scrum Professional®-ScrumMaster (CSP®-SM) certification. What you will Learn Learn to find practical solutions and improve your implementation of Scrum in the workplace. Aside from the pride gained and earning potential of attaining CSP® level, you can also: Attend exclusive CSP® events with other leaders in Scrum and Agile Attract more recruiters and command a higher rate of pay Establish a gateway and milestone toward becoming CST®, CEC, or CTC Receive a free premium subscription to the world's largest Agile assessment and continuous improvement platform, Comparative Agility® Hold an Advanced Certified ScrumMaster® (A-CSM®) certification with the Scrum Alliance · Attend a certified CSP®-SM educational offering to gain advanced Agile facilitation and coaching skills and learn progressive techniques in service to the Product Owner and the Development Team Successfully complete all educator-designed components of an approved educational offering. This may include pre- or post- course work as deemed necessary by your approved educator to complete the learning objectives. You will be asked to accept the CSP®-SM License Agreement and complete your Scrum Alliance membership profile Validate at least 24 months of work experience specific to the role of Scrum Master (from within the past five years)

Portfolio, Programs, and Project Offices (P3O) Foundation
By IIL Europe Ltd
Portfolio, Programme, and Project Offices (P3O®) Foundation P3O® is the AXELOS standard for the design of decision-making processes regarding changes in organizations. P3O provides a guideline for the design of portfolio, programme, and project offices in organizations. The P3O Foundation course is an interactive learning experience. The P3O Foundation-level content provides you with sufficient knowledge and understanding of the P3O guidance to interact effectively with, or act as an informed member of, an office within a P3O model. It enables you to successfully complete the associated P30 Foundation exam and achieve the qualification. In this course, you will be prepared to successfully attempt the P3O Foundation exam and learn how to implement or re-energize a P3O model in their own organization. What you will Learn Individuals certified at the P3O Foundation level will be able to: Define a high-level P3O model and its component offices List the component offices in a P3O model Differentiate between Portfolio, Programme, and Project Management List the key functions and services of a P3O List the reasons for establishing a P3O model Compare different types of P3O models List the factors that influence selection of the most appropriate P3O model for an organization Define the processes to implement or re-energize a P3O Benefits: Fast-track programme for those who want to achieve P3O Foundation qualification Practical case study and scenarios Attractive slides and course book Introduction to P3O What is the purpose of P3O? Definitions What are P3Os? Portfolio, programme, and project lifecycles Governance and the P3O Designing a P3O Model Factors that affect the design Design considerations What functions and services should the P3O offer? Roles and responsibilities Sizing and tailoring of the P3O model Introduction to P3O What is the purpose of P3O? Definitions What are P3Os? Portfolio, programme, and project lifecycles Governance and the P3O Designing a P3O Model Factors that affect the design Design considerations What functions and services should the P3O offer? Roles and responsibilities Sizing and tailoring of the P3O model Why Have a P3O? How a P3O adds value Maximizing that value Getting investment for the P3O Overcoming common barriers Timescales How to Implement or Re-Energize a P3O Implementation lifecycle for a permanent P3O Identify Define Deliver Close Implementation lifecycle for a temporary programme or project office Organizational context Definition and implementation Running Closing Recycling How to Operate a P3O Overview of tools and techniques Benefits of using standard tools and techniques Critical success factors P3O tools P3O techniques

100 Hours Yoga Teacher Training
By Kranti Yoga
Embark on a transformative journey with Kranti Yoga's 100-Hour Workshop, a soul-enriching experience designed to deepen your understanding of yoga, nurture self-discovery, and enhance overall well-being. Whether you're a seasoned practitioner or a novice, this immersive workshop invites you to explore the profound teachings of yoga in a supportive and tranquil environment.

A System Approach to Good Pharmacovigilance Practice
By Research Quality Association
Course Information In today's evolving landscape, pharmacovigilance (PV) systems face ongoing challenges due to global, national, and company-specific events. This course focuses on developing personnel equipped to navigate these complexities and improve the safe use of medicinal products. We emphasise continuous global thinking, communication, and strategic planning, ensuring adaptability across various levels. The course explores maintaining PV system functionality while adhering to regulatory requirements. Participants will apply European regulatory standards to enhance and sustain PV system effectiveness, contributing to improvement initiatives and ensuring operational integrity. The course covers PV system intricacies, regulatory compliance, and a 'systems approach' for auditors, quality assurance personnel, and PV practitioners. Through presentations and workshops, attendees gain insights into implementing and maintaining an effective PV system. Who should attend? Auditors Pharmacovigilance Quality System Managers Pharmacovigilance scientists The QPPV. Course benefits Throughout the course delegates will explore application of the legal requirements for the PV system and quality system and how to assure these systems. They will demonstrate their ability to contribute to: A systematic investigation of the pharmacovigilance system and its quality system Examination of how the pharmacovigilance system and quality system interact to achieve compliance. The risk-based approach to auditing the PV system and quality system The maintenance of 'inspection readiness' Explore how to investigate the complex PV system Discussions about how to monitor and maintain the PV system and assure compliance. Course Objectives Clarify what has to be done: Explore application of the legal requirements. Explore how to do what has to be done: Adopt a systemic approach to systematically investigate or implement and maintain the PV system and quality system Examine how a compliant PV system and a compliant quality system interact to achieve compliance with regulatory requirements for PV Explore how to investigate the complexity of the PV system. Discus how to identify what is missing or what needs to be improved: Discuss how to monitor and maintain the PV system and assure compliance. This course will assist delegates with: An understanding of key system principles, A practical approach to implementing, maintaining and monitoring the PV system and its quality system A procedure to share expertise to increase efficiency and confidence. This course is structured to encourage delegates to: Discuss and develop ideas - Share knowledge and experiences - Solve specific problems. By the end of the course delegates will be able to: Understand better the pharmacovigilance system, its quality system and how the components interact to achieve the objectives of pharmacovigilance Investigate, and analyse the pharmacovigilance system and to identify what is missing and what needs to be improved. Tutors Tutors will be comprised of (click the photos for biographies): Jana Hyankova Head of PV Department, IVIGEE Services a.s. Programme Please note timings may be subject to alteration. Day 1 08:30 Welcome, registration, course objectives and introduction to work groups Housekeeping notices, meet other delegates, explore how to work in your work group, course objectives. Clarify the definition and objectives of Pharmacovigilance. 09:30 The Regulatory Framework for Pharmacovigilance Identify the relevant regulations and directives. Explore GVP guidance, structure of the modules and standard format of each module. 10:00 The Pharmacovigilance System Exploration of how to organise what has to be done, communications. Exploration of the structures and processes for pharmacovigilance. 10:30 Break 11:00 Workshop 1 and Feedback Exploring an organisational model of the pharmaceutical company- cooperation between PV and other stakeholders. 12:00 The Quality System for pharmacovigilance Exploration of the structures, processes for the PV quality system and discussion of how it interacts with the pharmacovigilance system to meet the objectives of pharmacovigilance effectively and efficiently. 13:00 Lunch 14:00 Workshop 2 and Feedback The quality system puzzle. Explore the organisation of the PV quality system and how it interacts with the PV system. 14:30 The Quality System for pharmacovigilance Exploration of the structures processes for the PV quality system and discussion of how it interacts with the pharmacovigilance system to meet the objectives of pharmacovigilance effectively and efficiently. 15:00 Description of PV System 15:30 Break 15:30 Workshop 3 and Feedback The quality system puzzle Explore the organisation of the PV quality system and how it interacts with the PV system. 16:00 The Pharmacovigilance Safety Master File Construction of the Pharmacovigilance System Master File and its purpose. 17:00 Workshop 3 and Feedback Description of PV System. 18:00 End of Day Day 2 08:30 Drug Safety in the Clinical Trial Environment - Part 1 Information flow and responsibilities of the sponsor. 09:30 Workshop 4 and Feedback Drug Safety in the Clinical Trial environment: Information flow and responsibilities of the sponsor. 10:30 Break 11:00 Drug Safety in the Clinical Trial Environment - Part 2 Information flow and responsibilities of the sponsor. 12:00 Lunch 13:00 Workshop 5 and Feedback Drug Safety in the clinical trial environment: Information flow and responsibilities of the sponsor. 13:30 Processing of Safety Data Exploration of safety data processing, verification, validation, follow up, formatting and collation, reporting requirements, quality and data management. 15:00 Break 15:30 EudraVigilance Exploration of how EudraVigilance supports the PV system. 16:15 Signal Detection and Evaluation/Risk Benefit Assessment: Pharmacovigilance Risk Assessment Committee (PRAC): What is a signal? What are the regulatory requirements? How is signal detection and evaluation conducted? Qualitative and quantitative methods of signal detection. Risk benefit assessment. 17:00 Risk Management Plans A cornerstone of Pharmacovigilance safety communications, direct healthcare professional communication 18:00 End of Day Day 3 08:30 The Pharmacovigilance Risk Assessment Committee (PRAC) Exploration of how good practice is achieved. Composition, role and responsibilities. Examples of referrals. 09:15 Development Safety Update Reports (DSURs): Regulatory requirements, exploring good practice, report format, reference safety information, schedule of submission, analysis evaluations and distribution. 10:00 Periodic Safety Update Reports (PSURs)/Periodic Benefit Risk Evaluation Reports (PBRERs) Regulatory requirements, exploring good practice, report format, reference safety information, schedule of submission, analysis evaluations and distribution. 10:30 Break 11:00 Periodic Safety Update Reports (PSURs)/Periodic Benefit Risk Evaluation Reports (PBRERs) Regulatory requirements, exploring good practice, report format, reference safety information, schedule of submission, analysis evaluations and distribution. 12:00 Workshop 6 and Feedback To explore the compilation and submission of the PSUR. 13:00 Lunch 13:30 Role of the QPPV Exploration of the legal responsibilities of the QPPV and the MAH. 14:30 Break 15:00 Workshop 7 and Feedback To explore the challenges faced by the QPPV. 15:30 End of course Extra Information Face-to-Face Course Course material This course will be run completely online. You will receive an email with a link to our online system, which will house your licensed course materials and access to the remote event. Please note this course will run in UK timezone. The advantages of this include: Ability for delegates to keep material on a mobile device< Ability to review material at any time pre and post course Environmental benefits – less paper being used per course Access to an online course group to enhance networking You will need a stable internet connection, a microphone and a webcam. CPD Points 23 Points Development Level Develop

Introduction to Computer Systems Validation
By Research Quality Association
Course Information Join our comprehensive three-day training programme tailored for individuals seeking a foundational grasp of computerised system validation. This course equips participants with essential knowledge to effectively validate systems within their respective organisations for utilisation in GxP (GLP, GCP, GMP, GDP, and GPvP) environments. Attendees will also gain proficiency in auditing validated computerised systems, ensuring compliance with pertinent GxP regulations. Commencing with an overview of regulatory prerequisites and the system life cycle, the course transitions into practical aspects, centered around validating computerised systems and conducting subsequent audits. Engage in a dynamic blend of presentations, interactive discussions, and hands-on practical workshops throughout the course. This course will provide delegates with an understanding of the computerised system validation process, including: Definition of end user requirements Risk management, including supplier assessment and techniques for audit planning Validation planning and reporting Linking system development with good business practices Formal testing and qualification Understanding of data integrity and security issues How to assess system validation documentation to verify compliance. Is this course for you? IT professionals new to implementing computerised systems into regulated environments Quality professionals who monitor or audit computerised systems System owners, end users, tester and project staff. Tutors Tutors will be comprised of (click the photos for biographies): Nichola Stevens Director and Principal Consultant, Nuncius Compliance Solutions Ltd Barry McManus Consultancy Partner, Empowerment Quality Engineering Ltd Programme Please note timings may be subject to alteration. Day 1 09:00 Welcome, Introduction and Course Objectives 09:45 Why Validate? Regulations and Guidance on Computerised System Validation Overview of the regulations and guidance applicable to CSV and their key expectations. 10:30 Break 10:45 The System Lifecycle The concept of the SLC and the key outputs from it. 12:00 Lunch 12:45 The Validation Process The approach to validation for different system types and a look at some of the key deliverables. 14:00 Project Introduction 14:15 Exercise 1 - User Requirements Capturing, agreeing and documenting the user requirements for a system. 15:15 Break 15:30 Exercise 1 - Feedback 16:00 Risk Management Risk management and its impact on validation. Identifying the deliverables required. Then group discussion on risk assessment for three systems. 17:00 Questions and Answers Answers to any outstanding questions from Day 1. 17:15 Close of Day Day 2 09:00 Supplier Assessment The different approaches to supplier assessment and the things to be considered when assessing a supplier. 10:15 Exercise 2 - Supplier Assessment Planning a vendor audit with a focus on the key validation deliverables. 11:00 Break 11:15 Exercise 2 - Feedback 11:45 Test Overview and Test Planning The different test phases, the purpose of each test phase and things to be considered when planning and reporting testing. 12:45 Lunch 13:30 Test Overview and Test Planning Continued. 14:15 Test Script Design, Execution and Review What a good test script looks like and the key things to consider when creating, executing and reviewing a test script. 15:30 Break 15:45 Exercise 3 - Creating a Test Script Create a test script based on user requirements created on Day 1. 17:15 Close of Day Day 3 09:00 Exercise 3 Feedback 09:30 Infrastructure Configuration and Qualification 10:30 Break 10:45 Validation Reporting Overview of the Validation Report and what should be included in it. 11:15 Maintaining the Validated State The procedures and records needed to ensure the system remains fit for purpose. 12:30 Lunch 13:15 Change Control Key concepts related to making changes to validated systems. 14:00 Data Integrity and Security How can we assure the integrity and security of our data. 15:15 Break 15:30 Course Objectives Summary and Panel Discussion A round up of key learning from the course. 17:00 Close of Course Extra Information Face-to-face course Course Material Course material will be available in PDF format for delegates attending this course. The advantages of this include: Ability for delegates to keep material on a mobile device Ability to review material at any time pre and post course Environmental benefits – less paper being used per course. The material will be emailed in advance of the course and RQA will not be providing any printed copies of the course notes during the training itself. Delegates wishing to have a hard copy of the notes should print these in advance to bring with them. Alternatively delegates are welcome to bring along their own portable devices to view the material during the training sessions. Remote course Course Material This course will be run completely online. You will receive an email with a link to our online system, which will house your licensed course materials and access to the remote event. Please note this course will run in UK timezone. The advantages of this include: Ability for delegates to keep material on a mobile device Ability to review material at any time pre and post course Environmental benefits – less paper being used per course Access to an online course group to enhance networking. You will need a stable internet connection, a microphone and a webcam. CPD Points 19 Points Development Level Develop

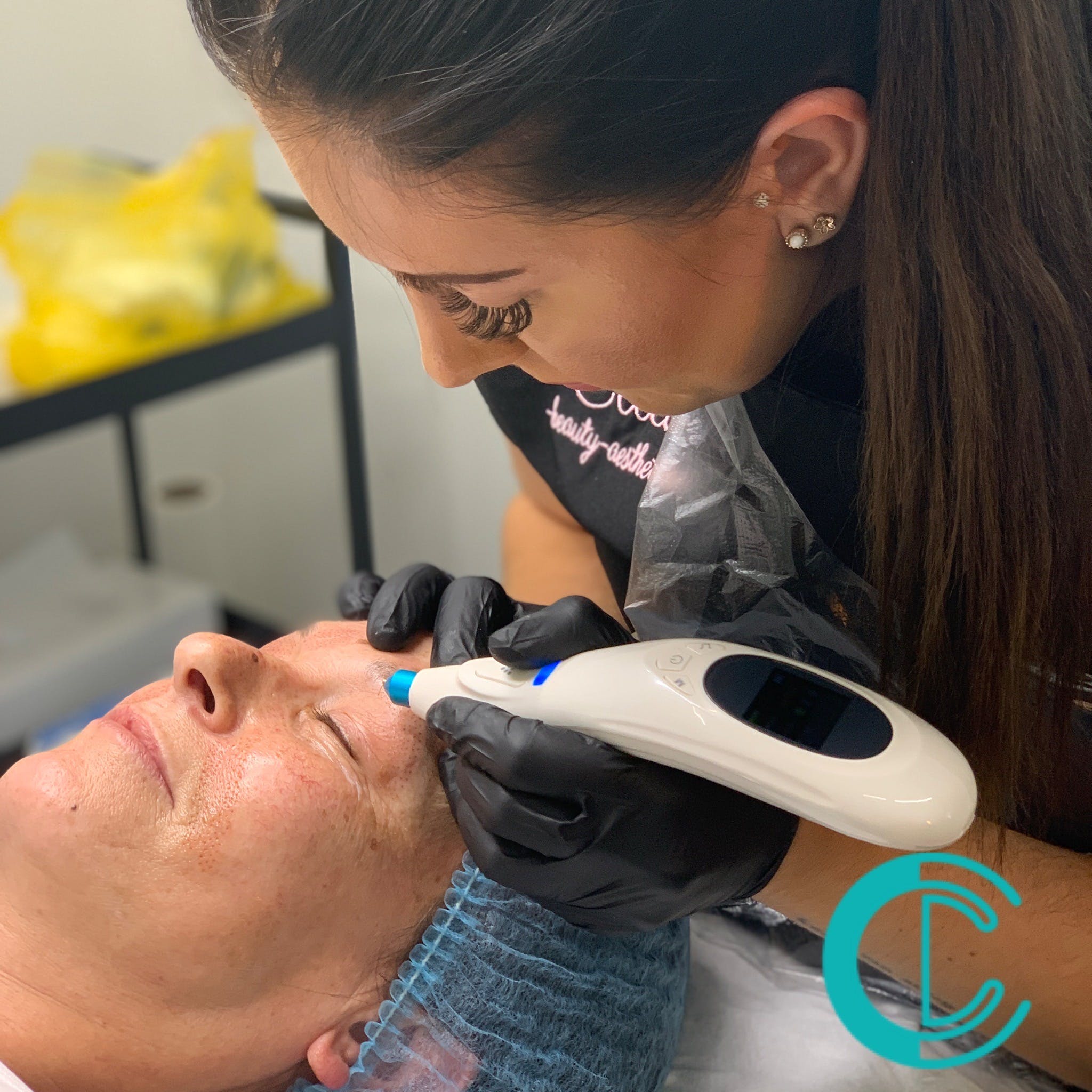
Plasma Fibroblast Skin Tightening Training
By Cosmetic College
Plasma skin tightening is a non-invasive precision technique that focuses on lifting and tightening loose skin on the face and body, with long-lasting results. Our plasma pen courses provide aesthetic practitioners with the ability to perform plasma pen treatments to a high standard; as the leading plasma pen training centre, we pride ourselves on training our practitioners to an extremely high standard to ensure their success. Our Plasma Fibroblast course has been structured from start to finish with you in mind to assure that when you complete your training, you will leave with the specialist skills that will help you feel confident in performing Plasma Fibroblast procedures. This intensive course includes theory study via our e-learning portal and practical training with model clients. All courses are kept small and focused, with a maximum of 6 learners in a class. Course prerequisites We accept students aged 18 and over. The student must have good written and spoken English. No prior experience or qualifications are required to enrol on this course. Course Structure Anatomy and physiology of the skin Infection control Sharps and hazardous waste training First aid and anaphylaxis training Pre-study plasma skin tightening theory Skin regeneration Skin tightening for face and neck lifting Reduce the appearance of stretch marks and scarring Full consultation process Faux skin peel practice Non-surgical skin tightening and lifting, including reduction of lines and wrinkles Practical training 1 model Clinical set up Professional live demonstrations Upgrade your training and include a medical-grade Beautier Plasma Pen with probes by Beautier Cosmetics for just 449 saving 550 Frequently Asked Questions How much is the course? Our Plasma Fibroblast training course is normally priced at 2,000. This course sometimes is available at a discounted rate subject to terms and conditions or specific promotional offers. How can I book? We have a few options for you to book. You can book by selecting an available training date above here on our website, by contacting us through email at hello@cosmetic.college or by contacting us on 0333 015 5117. What qualifications do I need to be able to enrol on this course? No previous experience or qualifications are required for you to enrol onto this course. Whether you are a beauty pro, aesthetician, permanent makeup artist or a complete novice, we will give you all the training you need to become a successful Plasma Fibroblast practitioner. What is the course duration? 1 day + pre-study via our online learning platform. Is there a kit included in the price? This course does not include a kit in the training fees however you can upgrade your training and include a medical-grade Beautier Professional Plasma Pen for 449 using our partnership discount exclusively for Cosmetic College students. Which accreditations does this course have? CPD Accreditation Group Certification provided on successful completion of the course Is a deposit required to book? All enrolments are charged an administration fee which is non-refundable. When you enrol you can elect to pay a deposit of 10% plus the administration fee or pay the total training course in full. We have full details of the terms and conditions of training course enrolments here Where is the Cosmetic College The Cosmetic College is located at: 3 Locks Court, 429 Crofton Road, Orpington, BR6 8NL
Hydrogen - Technology, Economics and Business Cases
By EnergyEdge - Training for a Sustainable Energy Future
About this Virtual Instructor Led Training (VILT) Asia Pacific is set to be the largest and fastest growing Hydrogen market globally. This growth is driven by decarbonisation of energy-use, ammonia production and rising demand of fuel cell electric vehicles. Hydrogen as a fuel has outstanding energy carrying capacity and many application possibilities ranging from Petroleum refinery, Ammonia and Methanol production, Transportation and Power generation. Although the current petrochemical market segment will remain strongest in the near future, it is the transport and power sector which spurs the vision of a massive market takeoff in the next decade. The ever-rising share of renewable energies require flexible and scalable storage solutions, which in turn offers many additional revenue streams beyond pure electricity sales. Adding to this the strong impetus towards decarbonization of the transport sector from cars, trucks, trains to ships and even airplanes creates the breed for an exciting and yet untapped market potential. This course aims to clarify and assess the hydrogen business case along its value chain and versatile market applications. Training Objectives Understanding current hydrogen market status and recent developments Major drivers and inhibitors influencing the growth of the market Understanding and comparing various production technology processes Challenges and solutions in transport, distribution and storage of hydrogen Mapping the many petrochemical, energy and transport applications Analyse business cases from around the world and understand their economics Target Audience Project developers Equipment Manufacturers Oil, Gas and Petrochemical sector companies IPPs and utilities Transport sector companies and port operators Policy makers and regulators Investors and lenders Course Level Basic or Foundation Training Methods The VILT will be delivered online in 4 half-day sessions comprising 4 hours per day, including time for lectures, discussion, quizzes and short classroom exercises. Additionally, some self-study will be requested. Participants are invited but not obliged to bring a short presentation (10mins max) on a practical problem they encountered in their work. This will then be explained and discussed during the VILT. A short test or quiz will be held at the end the course. Trainer Your expert course leader is an internationally renowned energy communicator and business educator, focused on the interconnected clean energy transition topics of renewable power, energy storage, energy system electrification and hydrogen. His own independent technology tracking, market assessment and opportunity/risk analysis is delivered to clients through a mix of business advisory work, commissioned content, small-group training (online & in-person), and one-to-one executive coaching (online). In the hydrogen sector, he is currently lead consultant and trainer to the World Hydrogen Leaders network, and writer of their 'This Week in Hydrogen' news column. He is also co-presenter of the 'New Energy Chinwag' podcast, which regularly covers hydrogen-related issues. During more than 15 years as an independent energy expert, he has helped companies from large multinationals to innovative start-ups - totalling assignments in over 30 countries across 5 continents. Most recently, he has presented clean energy training in locations as diverse as Singapore, the UK, South Africa, The Philippines, the USA, Mexico, Spain and Dubai - and, in recent times of course, online to international audiences from across the world. Prior to this, he was Research Director for over 10 years at Informa, a $9 billion business intelligence provider; where he drove new market identification, analysis and project deployment work, and managed teams in the UK and US. He has a strong science background, holding a 1st Class Honours degree in Natural Sciences from the University of Cambridge, a PhD in Earth Sciences and a further Diploma in Economics & Sustainability from the UK's Open University. POST TRAINING COACHING SUPPORT (OPTIONAL) To further optimise your learning experience from our courses, we also offer individualized 'One to One' coaching support for 2 hours post training. We can help improve your competence in your chosen area of interest, based on your learning needs and available hours. This is a great opportunity to improve your capability and confidence in a particular area of expertise. It will be delivered over a secure video conference call by one of our senior trainers. They will work with you to create a tailor-made coaching program that will help you achieve your goals faster. Request for further information about post training coaching support and fees applicable for this. Accreditions And Affliations

Advanced Level - Assessing and Monitoring the Condition of Power Transformers by Oil Analysis
By EnergyEdge - Training for a Sustainable Energy Future
About this Virtual Instructor Led Training (VILT) The objective of this 4-half-day Virtual Instructor Led Training (VILT) course is to equip participants with the required knowledge so that they can define and implement an effective oil analysis programme, monitor the condition of transformers and understand oil analysis reports as part of their maintenance strategy. This VILT course is delivered in partnership with ENGIE Laborelec. Training Objectives Participants will be able to go through the following areas during the VILT course: Condition monitoring of power transformers through oil analyses: Transformer Health Index, Transformer Fleet Condition Assessment, Risk Matrix Best practices Which oil analyses/how frequent DGA: focus on partial discharges, sparking, stray gassing, catalytic effects / interpretation systems. Interpretation of results Why and when to perform electrical measurements? International standards: IEEE, IEC, Duval, ASTM, etc Maintenance guidelines Trending and reporting Sampling techniques Use of online monitoring for DGA/water: implementation and real-case advantages etc: New developments: importance of methanol as ageing marker, temperature correction of ageing markers, stray gassing of oils in service etc REX, case studies and real-case exercises concerning power transformers within generation and high-voltage grids Discussion of reports on power transformers Use of unused insulating transformer oils Market trends in the use of inhibited and uninhibited oils Differences, advantages and drawbacks of uninhibited compared with inhibited oils Importance of additives, oxidation stability tests, stray gassing, etc Target Audience The VILT course is intended for a wide audience, including professionals who are maintenance staff, electrical engineers and asset managers responsible for transformers. Typical industries are powerplants, high voltage grids or large industrial sites. Course Level Basic or Foundation Training Methods The VILT course will be delivered online in 4 half-day sessions comprising 4 hours per day, with 2 x 10 minutes break per day, including time for lectures, discussion, quizzes and short classroom exercises. Course Duration: 4 half-day sessions, 4 hours per session (16 hours in total). This VILT course is delivered in partnership with ENGIE Laborelec. Trainer Your expert course leader is a senior expert in the field of insulating and lubricating oils and of condition monitoring of power transformers and turbines. He has 20 years of experience within power industry and since 2010 Key Expert within the ENGIE group. He has worked on various international projects such as the Al Dur power plant in Bahrain, Shuweihat II power plant in UAE, PP11 and Jubail Marafiq power plants in Saudi Arabia and Sohar II and Barka III power plants in Oman. He has also been involved in consulting work in Malaysia for Hyrax Oil. Other assignments include Shell, Nynas, Total, ABB, Alstom, CG Pauwels in Europe. POST TRAINING COACHING SUPPORT (OPTIONAL) To further optimise your learning experience from our courses, we also offer individualized 'One to One' coaching support for 2 hours post training. We can help improve your competence in your chosen area of interest, based on your learning needs and available hours. This is a great opportunity to improve your capability and confidence in a particular area of expertise. It will be delivered over a secure video conference call by one of our senior trainers. They will work with you to create a tailor-made coaching program that will help you achieve your goals faster. Request for further information about post training coaching support and fees applicable for this. Accreditions And Affliations

Technical Report Writing and Presentation Skills for Oil & Gas Engineers and Technical Professionals
By EnergyEdge - Training for a Sustainable Energy Future
About this Training Course Oil & Gas professionals increasingly need to translate complex findings, analysis and recommendations for effective decision-making. If you face challenges in getting your findings into paper, you will benefit from this training course. The Technical Report Writing and Presentation Skills for Oil & Gas Engineers and Technical Professionals course focuses on the unique needs of technical professionals who write for both technical and non-technical readers. This separately bookable training course will demonstrate how technical professionals can use their technical knowledge and logical edge to write in a reader-friendly style, produce grammatically accurate reports and persuasively communicate for buy-in purposes. An ILM Recognised programme. Training Objectives LEARNING OUTCOMES FOR TECHNICAL REPORT WRITING SKILLS By the end of this course, participants will be able to: Write and turn-around accurate technical documents quickly to meet deadlines and productivity goals Understand how technical reports should be structured by applying a systematic approach to the writing task, involving planning, drafting, revision and production Use clear & powerful language to target and persuade readers for positive results Use tried and tested proof reading techniques to check and review documents more effectively Identify and avoid common pitfalls in technical report writing LEARNING OUTCOMES FOR TECHNICAL PRESENTATION SKILLS By the end of this course, participants will be able to: Present and sell your technical presentation more effectively both internally and externally Plan and structure your meetings and presentations for maximum effect Capture your technical and non-technical audience by delivering your presentation with power and authority Handle questions, interruptions and objections with confidence Target Audience This programme has been researched and developed for all Oil & Gas Engineers and Technical Professionals Course Level Basic or Foundation Trainer Your course leader managed the Business Skills Unit of the British Council programmes as well as oversee all training-related matters from 1997. The trainer considers himself a trainer first and specialises predominantly in high-level writing and technical report writing programmes. In addition to conducting training in Technical Writing, the trainer has also been the chief editor for many large writing projects. These include a year-long project editing the entire Start-Up Manual (including the Black Start Manual) for Nippon Oil's Helang Integrated Platform, a 4-month project rewriting the manuals for the Puteri Dulang FSO off Terengganu, and editing the current revision of the PETRONAS Procedures and Guidelines for Upstream Activities (PPGUA). POST TRAINING COACHING SUPPORT (OPTIONAL) To further optimise your learning experience from our courses, we also offer individualized 'One to One' coaching support for 2 hours post training. We can help improve your competence in your chosen area of interest, based on your learning needs and available hours. This is a great opportunity to improve your capability and confidence in a particular area of expertise. It will be delivered over a secure video conference call by one of our senior trainers. They will work with you to create a tailor-made coaching program that will help you achieve your goals faster. Request for further information post training support and fees applicable Accreditions And Affliations

HV & MV Power System Design, Protection & Coordination - Virtual Instructor-Led Training (VILT)
By EnergyEdge - Training for a Sustainable Energy Future
Enhance your expertise in HV/MV power system design and protection coordination with EnergyEdge's virtual instructor-led training. Join now!

Search By Location
- MA Courses in London
- MA Courses in Birmingham
- MA Courses in Glasgow
- MA Courses in Liverpool
- MA Courses in Bristol
- MA Courses in Manchester
- MA Courses in Sheffield
- MA Courses in Leeds
- MA Courses in Edinburgh
- MA Courses in Leicester
- MA Courses in Coventry
- MA Courses in Bradford
- MA Courses in Cardiff
- MA Courses in Belfast
- MA Courses in Nottingham